Chapter 17: Q32E (page 567)
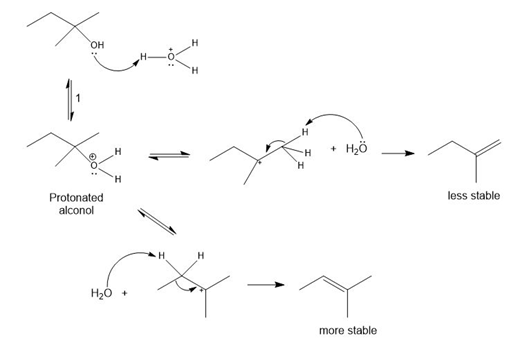
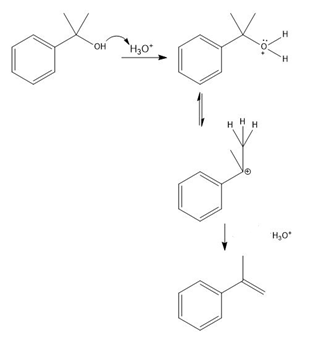
The conversion of 3° alcohols into alkenes under acidic conditions involves two cationic intermediates. For each reaction, draw the complete mechanism using curved arrows.
(a)
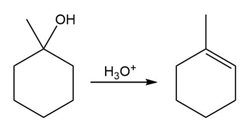
(b)
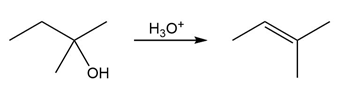
(c)
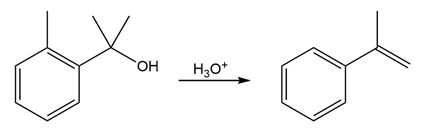
Short Answer
(a)
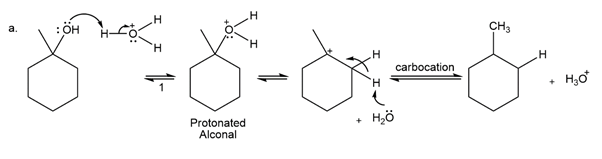
(b)
(c)
Learning Materials
Features
Discover
Chapter 17: Q32E (page 567)
The conversion of 3° alcohols into alkenes under acidic conditions involves two cationic intermediates. For each reaction, draw the complete mechanism using curved arrows.
(a)

(b)

(c)

(a)

(b)

(c)

All the tools & learning materials you need for study success - in one app.
Get started for free
Predict the product from reaction of the following substance (reddish brown=Br) with:
(a)
(b) Aqueous
(c)
(d)Dess-Martin periodinane
(e)
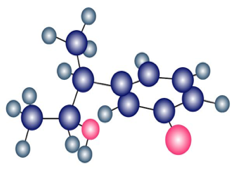
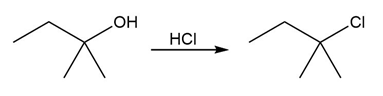
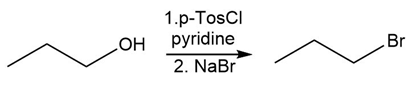
Question:Identify the type of substitution mechanism ,involved in the conversion of the alcohol shown into the corresponding alkyl halide.
a.
b)
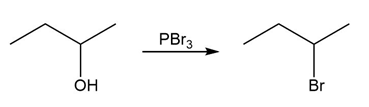
c)
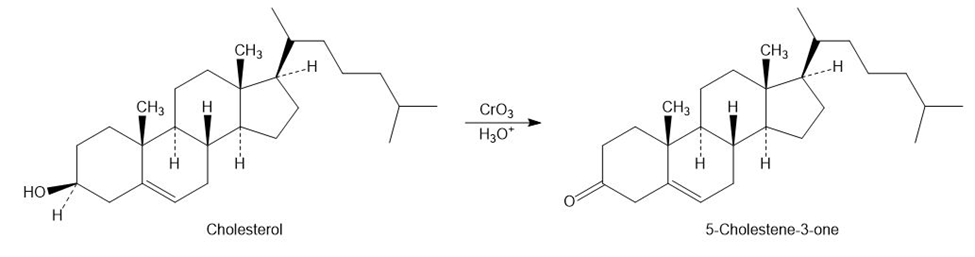
Assume that you need to prepare 5-Cholesten-3-one from Cholesterol. How could you use IR spectroscopy to tell whether the reaction was successful? What differences would you look for in the IR spectra of starting material and product?
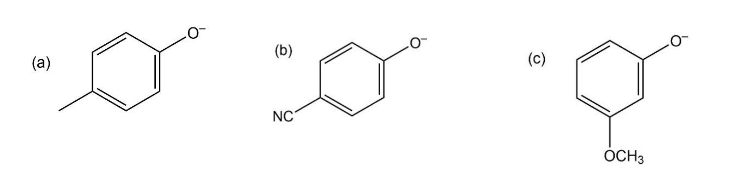
Phenols generally have lower pKa’s than aliphatic alcohols because of resonance stabilization with the aromatic ring. Draw all of the resonance contributors for the phenolate ions below. Make note of how the substituents either stabilize or destabilize the system.
A compound of unknown structure gave the following spectroscopic data:
Mass spectrum:
IR:
NMR: 1.4 (2H, quartet, J=7Hz); 1.2 (6H, singlet); 1.0 (1H, singlet); 0.9 (3H, triplet, J=7Hz)
NMR: 74, 35, 27, 25
(a) Assuming that the compound contains C and H but may or may not contain O, give three possible molecular formulas.
(b) How many protons (H) does the compound contain?
(c) What functional group(s) does the compound contain?
(d) How many carbons does the compound contain?
(e) What is the molecular formula of the compound?
(f) What is the structure of the compound?
(g) Assign peaks in the molecule’s NMR spectrum corresponding to specific protons.
What do you think about this solution?
We value your feedback to improve our textbook solutions.