Chapter 17: Q 32 E (page 567)
Question:The conversion of 3° alcohols into alkenes under acidic conditions involves two cationic intermediates. For each reaction, draw the complete mechanism using curved arrows.
a)
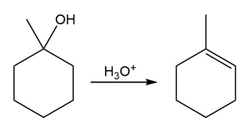
b)
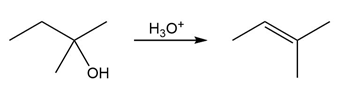
c)
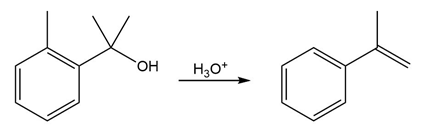
Short Answer
a)
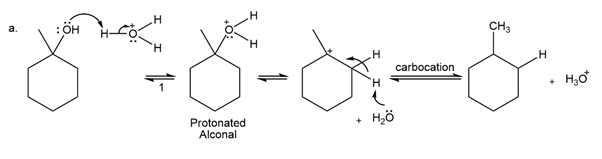
b)
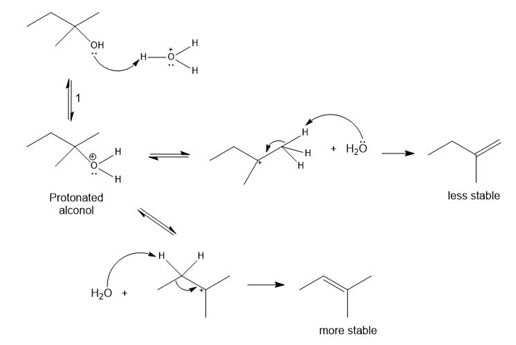
c)
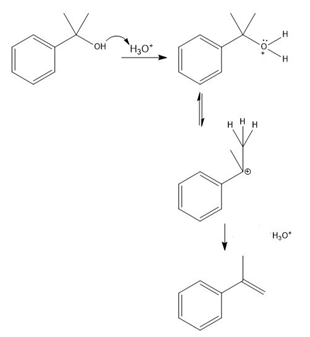
Learning Materials
Features
Discover
Chapter 17: Q 32 E (page 567)
Question:The conversion of 3° alcohols into alkenes under acidic conditions involves two cationic intermediates. For each reaction, draw the complete mechanism using curved arrows.
a)

b)

c)

a)

b)

c)

All the tools & learning materials you need for study success - in one app.
Get started for free
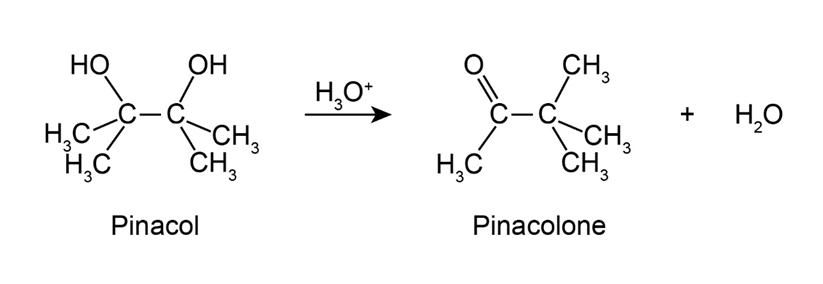
2,3-Dimethyl-2,3-butanediol has the common name pinacol. On heating with aqueous acid, pinacol rearranges to pinacolone, 3,3-dimethyl-2-butanone. Suggest a mechanism for this reaction.
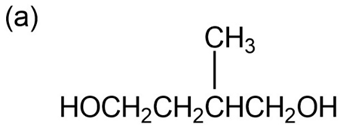
Question: Give IUPAC names for the following compounds:
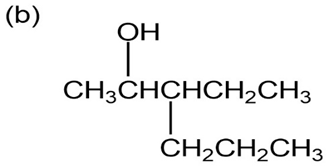
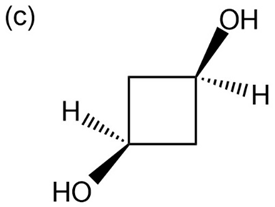
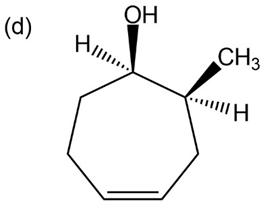
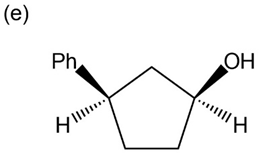
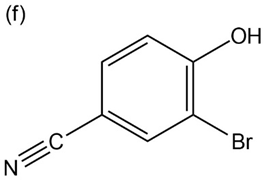
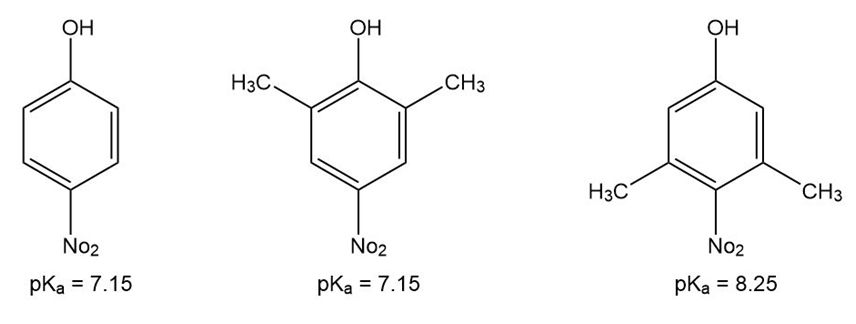
p-Nitrophenol and 2,6-dimethyl-4-nitrophenol both have pKa 5 7.15, but 3,5-dimethyl-4-nitrophenol has pKa 5 8.25. Why is 3,5-dimethyl-4- nitrophenol so much less acidic?
What products would you obtain from reaction of 1-pentanol with thefollowing reagents?
(a)
(b)
(c)
(d) Dess- Martin periodinane
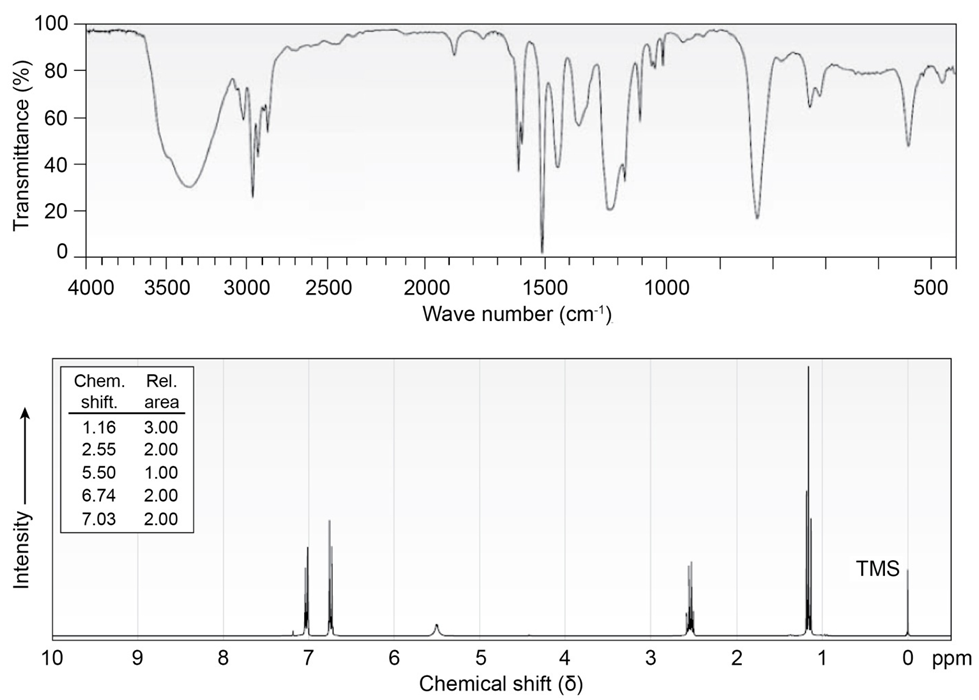
Compound A, , has the IR and NMR spectra shown. Proposea structure consistent with the observed spectra, and label each peak inthe NMR spectrum. Note that the absorption at disappears when is added.
What do you think about this solution?
We value your feedback to improve our textbook solutions.