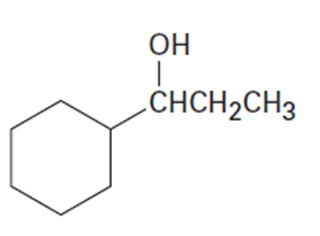
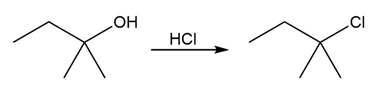
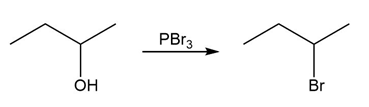
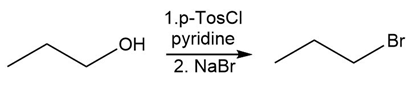
Chapter 17: Q17-68E (page 567)
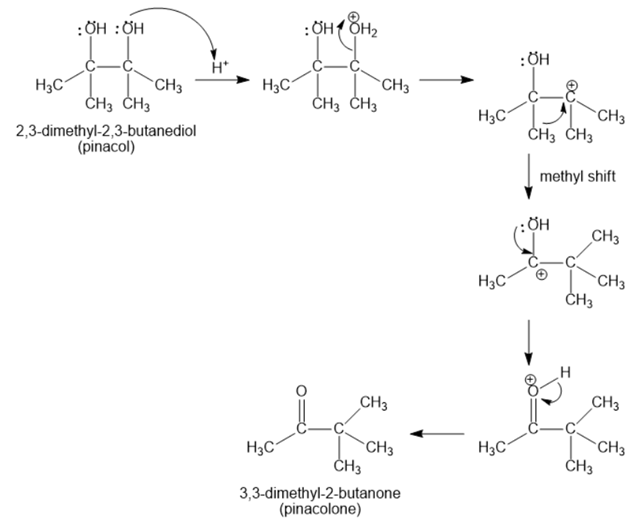
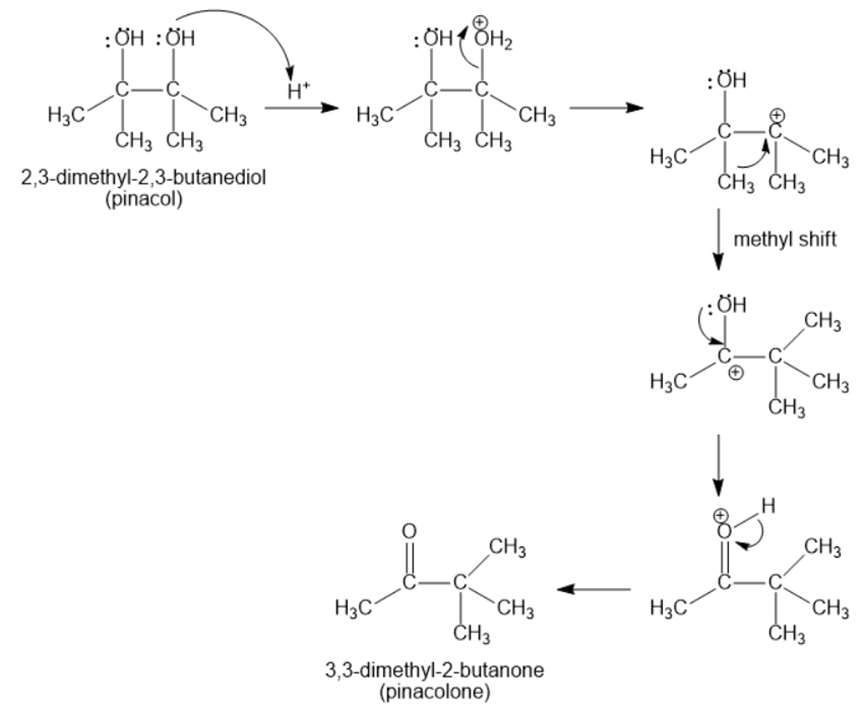
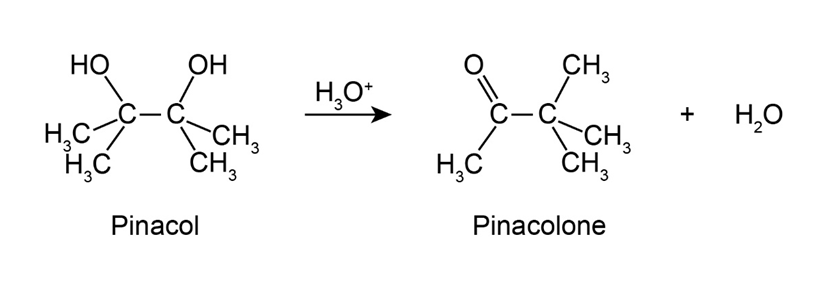
2,3-Dimethyl-2,3-butanediol has the common name pinacol. On heating with aqueous acid, pinacol rearranges to pinacolone, 3,3-dimethyl-2-butanone. Suggest a mechanism for this reaction.
Short Answer
The mechanism of 2,3-dimethyl-2,3-butanediol (pinacol) rearranges to 3,3-dimethyl-2-butanone (pinacolone) as;