Chapter 17: Q17-67E (page 567)
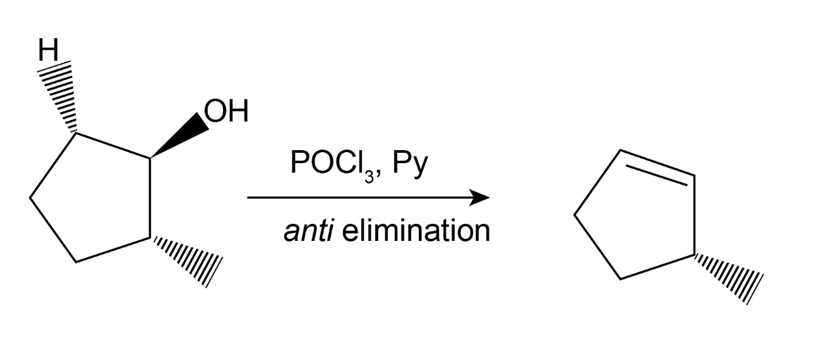
Dehydration of trans-2-methylcyclopentanol with POCl3 in pyridine yields predominantly 3-methylcyclopentene. Is the stereochemistry of this dehydration syn or anti? Can you suggest a reason for formation of the observed product? (Make molecular models!)
Short Answer
The reaction is as follows:
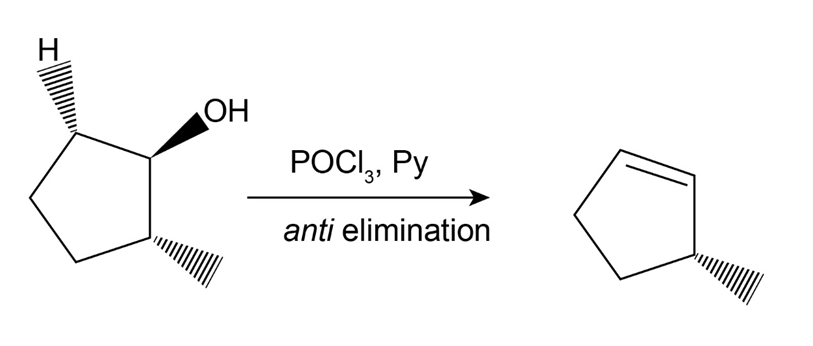
If the molecule undergoes syn elimination then the proton at the second carbon is removed and forms a 1-methyl-1-cyclopentene.
But in the reaction, 3-methylcyclopentene is formed. Therefore, the compound undergoes anti-elimination.
The activation energy of a syn elimination is too high when compared to an anti-elimination and thus the anti-elimination reaction is favored in spite of the less stable product.