Chapter 17: Q31E (page 567)
Identify the type of substitution mechanism involved in the conversion of the alcohol shown into the corresponding alkyl halide.
(a)
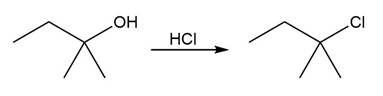
(b)
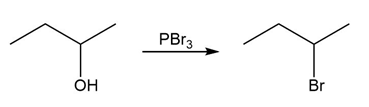
(c)

Short Answer
(a)
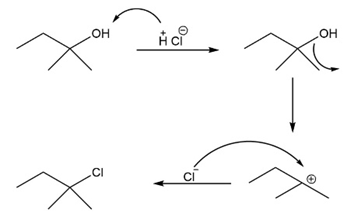
(b)

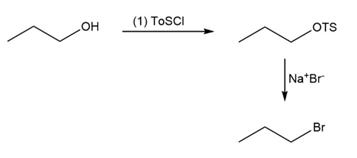
Learning Materials
Features
Discover
Chapter 17: Q31E (page 567)
Identify the type of substitution mechanism involved in the conversion of the alcohol shown into the corresponding alkyl halide.
(a)

(b)

(c)

(a)

(b)


All the tools & learning materials you need for study success - in one app.
Get started for free
How would you carry out the following transformations?
(a)

(b)
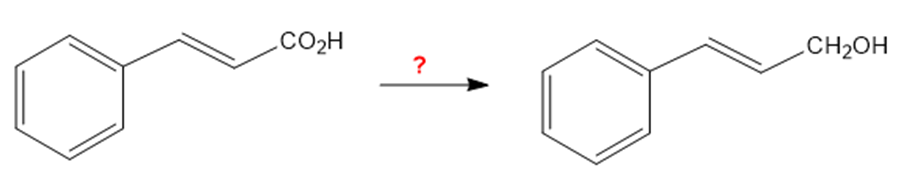
(c)

Question:Reaction of (S)-3-methyl-2-pentanone with methyl magnesium bromide followed by acidification yields 2,3-dimethyl-2-pentanol. What is the stereochemistry of the product? Is the product optically active?

Draw and name the eight isomeric alcohols with the formula C5H12O
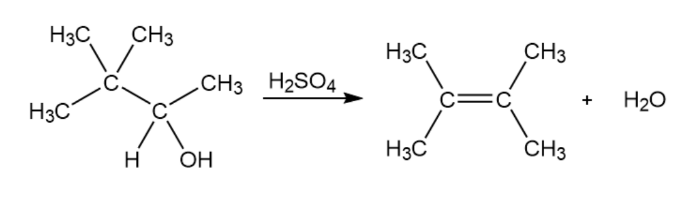
Question: Evidence for the intermediate carbocations in the acid-catalyzed dehydration of alcohols comes from the observation that rearrangements sometimes occur. Propose a mechanism to account for the formation of 2,3-dimethyl-2-butene from 3,3-dimethyl-2-butanol.
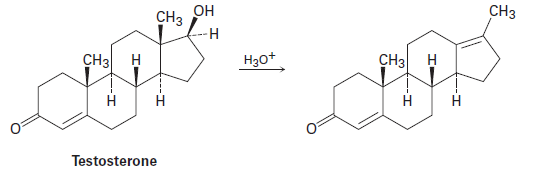
Testosterone is one of the most important male steroid hormones. When testosterone is dehydrated by treatment with acid, rearrangement occurs to yield the product shown. Propose a mechanism to account for this reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.