Chapter 17: Q38E (page 567)
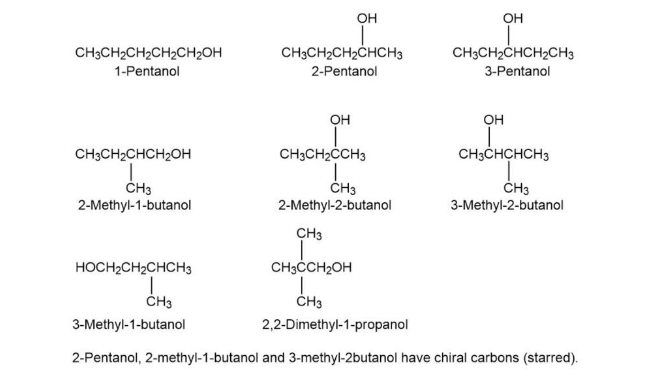
Draw and name the eight isomeric alcohols with the formula C5H12O
Short Answer
Isomers followed as:
Learning Materials
Features
Discover
Chapter 17: Q38E (page 567)
Draw and name the eight isomeric alcohols with the formula C5H12O
Isomers followed as:

All the tools & learning materials you need for study success - in one app.
Get started for free
What alcohols would give the following products on oxidation?
(a)
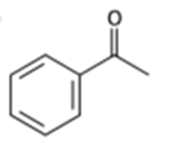
(b)
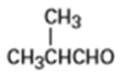
(c)
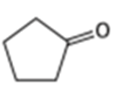
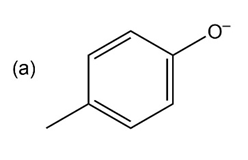
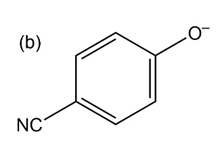
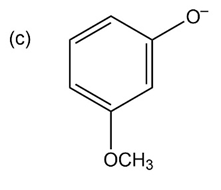
Question:Phenols generally have lower pKa’s than aliphatic alcohols because of resonance stabilization with the aromatic ring. Draw all of the resonance contributors for the phenolate ions below. Make note of how the substituents either stabilize or destabilize the system.
What products would you expect from oxidation of the following compounds with in aqueous acid? With the Dess-Martin periodinane?
The trimethylsilyl (TMS) protecting group is one of several silicon protecting groups for alcohols. For each reaction, draw the mechanism for the protection of (R)-3-bromo-1-butanol with the following silyl chlorides, using triethylamine as the base:
(a) tert-butyldimethylsilyl chloride (TBS-Cl)
(b) triisopropylsilyl chloride (TIPS-Cl)
(c) triethylsilyl chloride (TES-Cl)
How would you prepare the following compounds from 1-phenylethanol?
More than one step may be required.
(a) Acetophenone
(b) Benzyl alcohol
(c) m-Bromobenzoic acid
(d) 2-Phenyl-2-propanol
What do you think about this solution?
We value your feedback to improve our textbook solutions.