Chapter 17: Q 39 E (page 567)
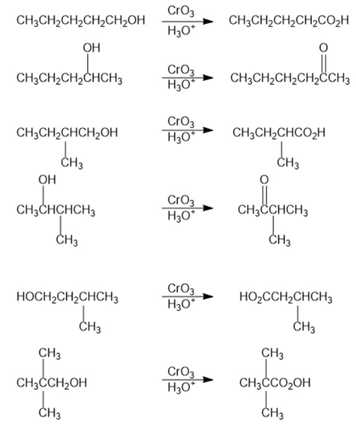
Question:Which of the eight alcohols that you identified in Problem 17-38 react with in aqueous acid? Show the products you would expect from each reaction
Short Answer
The reaction followed as:
Learning Materials
Features
Discover
Chapter 17: Q 39 E (page 567)
Question:Which of the eight alcohols that you identified in Problem 17-38 react with in aqueous acid? Show the products you would expect from each reaction
The reaction followed as:

All the tools & learning materials you need for study success - in one app.
Get started for free
How would you prepare the following substances from cyclopentanol?
More than one step may be required.
(a) Cyclopentanone
(b) Cyclopentene
(c) 1-Methylcyclopentanol
(d) trans-2-Methylcyclopentanol
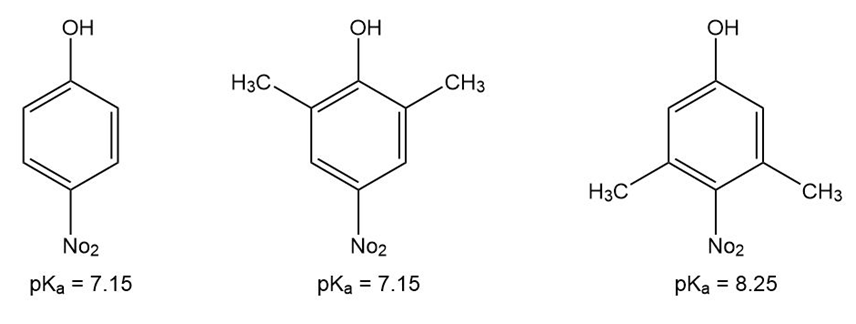
p-Nitrophenol and 2,6-dimethyl-4-nitrophenol both have pKa 5 7.15, but 3,5-dimethyl-4-nitrophenol has pKa 5 8.25. Why is 3,5-dimethyl-4- nitrophenol so much less acidic?
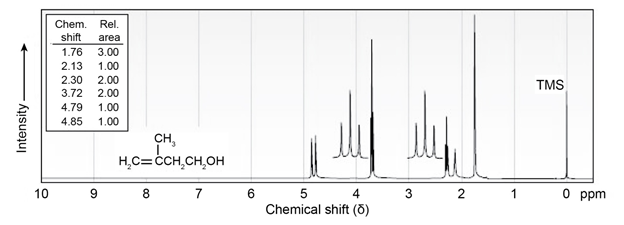
The NMR spectrum shown is that of 3-methyl-3-buten-1-ol. Assign
all the observed resonance peaks to specific protons, and account for
the splitting patterns.
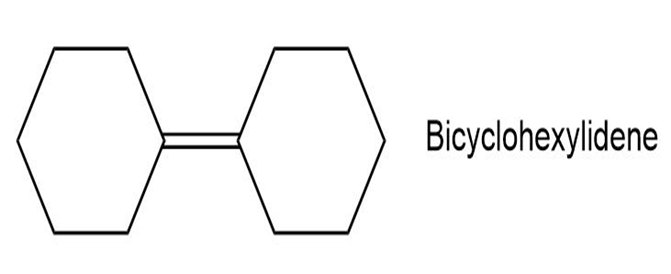
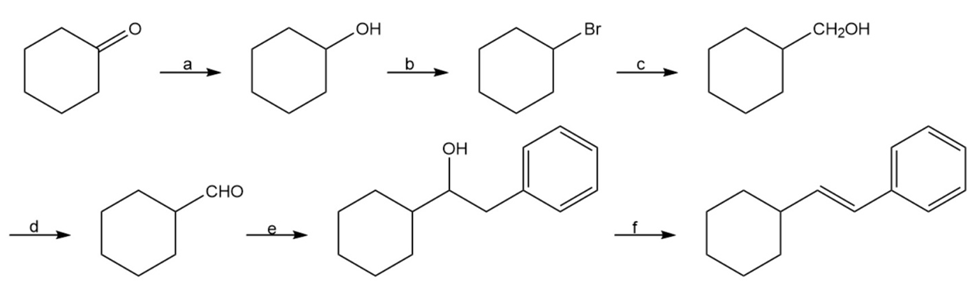
Propose a synthesis of bicyclohexylidene, starting from cyclohexanone as the only source of carbon.
Identify the reagents a–f in the following scheme:
What do you think about this solution?
We value your feedback to improve our textbook solutions.