(a)Cyclopentanone can be prepared from cyclopentanol by reacting cyclopentanol with and . The reaction can be given as:
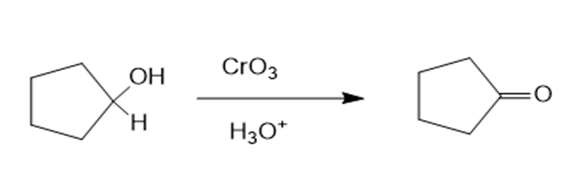
Reaction (a)
(b)Cyclopentene can be prepared from cyclopentanone by reacting cyclopentanol with and pyridine. The reaction can be given as:
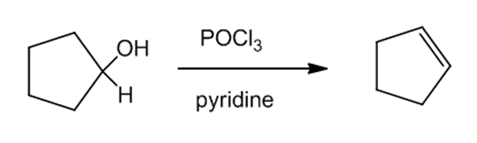
Reaction (b)
(c)1-Methylcyclopentanol can be prepared from cyclopentanol by reacting cyclopentanol in the presence of and to form cyclopentanone. This compound further reacts with and to form 1-Methylcyclopentanol. The reaction can be given as:
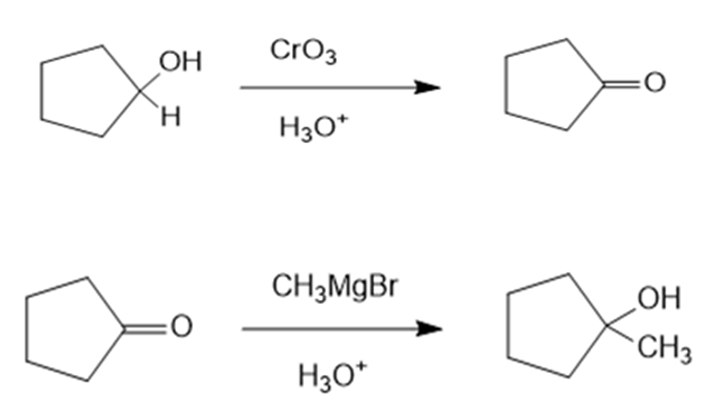
Reaction (c)
(d)The compound, trans-2-Methylcyclopentanol can be formed by reacting 1-Methylcyclopentanol with to form cyclopentene with methyl group as the substituent. This is further treated with , THF, and to form trans-2-Methylcyclopentanol. The reaction can be given as:
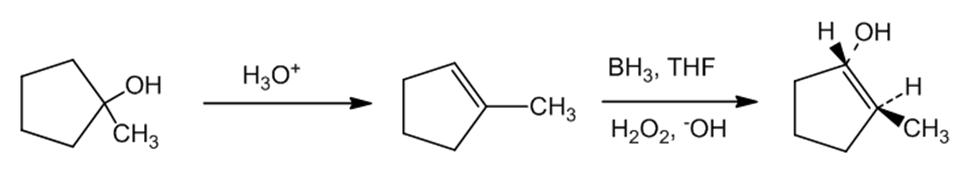
Reaction (d)













