Chapter 17: Q48E (page 567)
How would you prepare the following compounds from 1-phenylethanol?
More than one step may be required.
(a) Acetophenone
(b) Benzyl alcohol
(c) m-Bromobenzoic acid
(d) 2-Phenyl-2-propanol
Short Answer
(a)
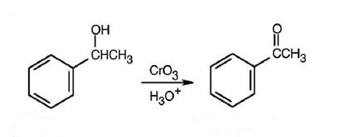
(b)
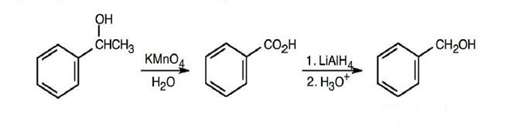
(c)
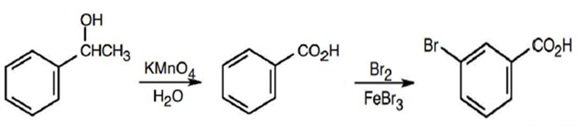
(d)
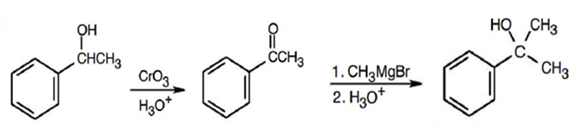
Learning Materials
Features
Discover
Chapter 17: Q48E (page 567)
How would you prepare the following compounds from 1-phenylethanol?
More than one step may be required.
(a) Acetophenone
(b) Benzyl alcohol
(c) m-Bromobenzoic acid
(d) 2-Phenyl-2-propanol
(a)

(b)

(c)

(d)

All the tools & learning materials you need for study success - in one app.
Get started for free
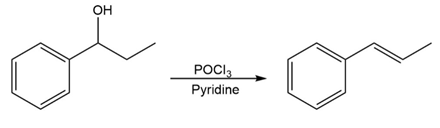
Question:For each reaction, write the mechanism using curved arrows for the conversion of the alcohol into the corresponding alkene with POCl3. In each case, explain the regiochemistry of the elimination.
a)
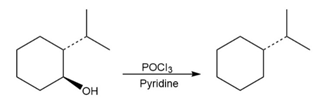
b)
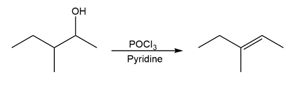
c)
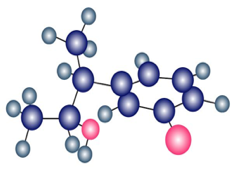
Predict the product from reaction of the following substance (reddish brown=Br) with:
(a)
(b) Aqueous
(c)
(d)Dess-Martin periodinane
(e)
Treatment of the following epoxide with aqueous acid produces a carbocation intermediate that reacts with water to give a diol product. Show the structure of the carbocation, and propose a mechanism for the second step.
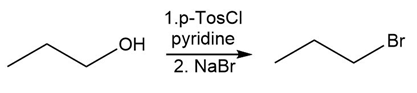
Identify the type of substitution mechanism involved in the conversion of the alcohol shown into the corresponding alkyl halide.
(a)
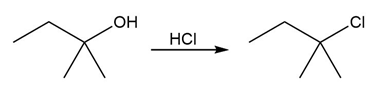
(b)
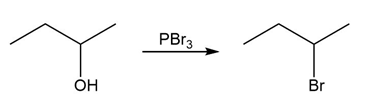
(c)
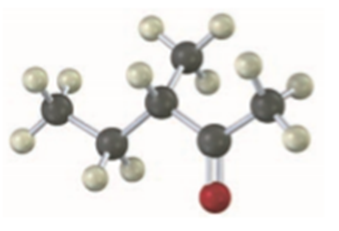
Name and assign R or S stereochemistry to the product (s) you would obtain by reaction of the following substance with ethylmagnesium bromide. Is the product chiral? Is it optically active? Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.