Chapter 17: Q17-70E (page 567)
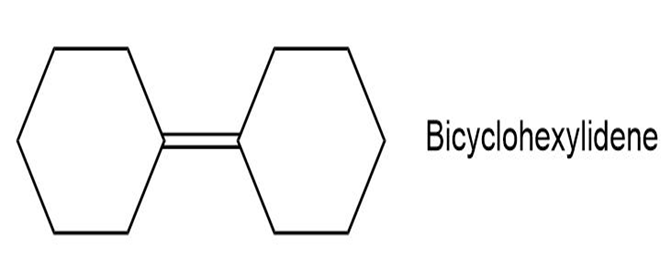
Propose a synthesis of bicyclohexylidene, starting from cyclohexanone as the only source of carbon.
Short Answer
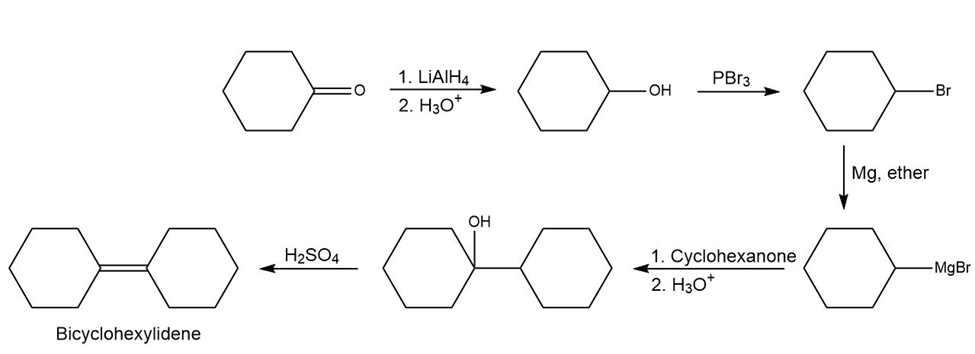
A synthesis of bicyclohexylidene from cyclohexanone is,
Learning Materials
Features
Discover
Chapter 17: Q17-70E (page 567)
Propose a synthesis of bicyclohexylidene, starting from cyclohexanone as the only source of carbon.

A synthesis of bicyclohexylidene from cyclohexanone is,

All the tools & learning materials you need for study success - in one app.
Get started for free
As a rule, axial alcohols oxidize somewhat faster than equatorial alcohols. Which would you expect to oxidize faster, cis-4-tert-butylcyclohexanol or trans-4-tert-butylcyclohexanol? Draw the more stable chair conformation of each molecule.
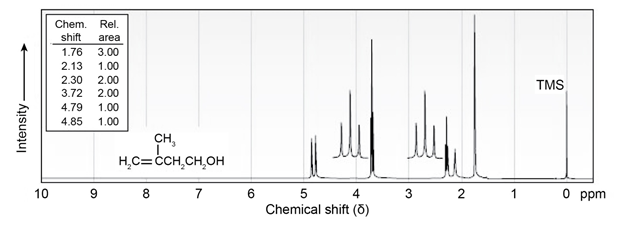
The NMR spectrum shown is that of 3-methyl-3-buten-1-ol. Assign
all the observed resonance peaks to specific protons, and account for
the splitting patterns.
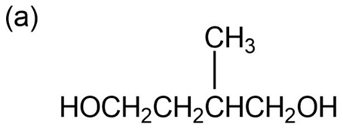
Question: Give IUPAC names for the following compounds:
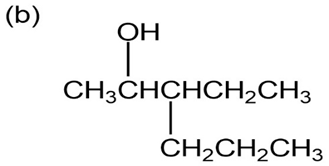
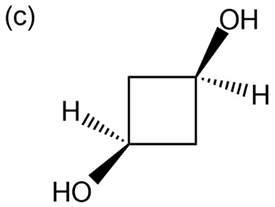
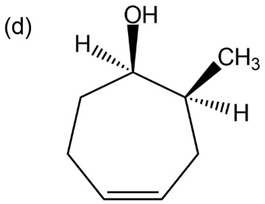
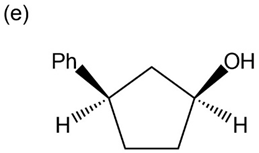
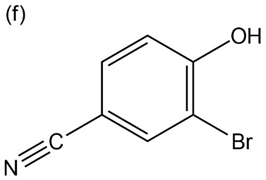
What products would you obtain from reaction of 1-pentanol with the
following reagents?
(a)
(b)
(c) , ,
(d) Dess–Martin periodinane
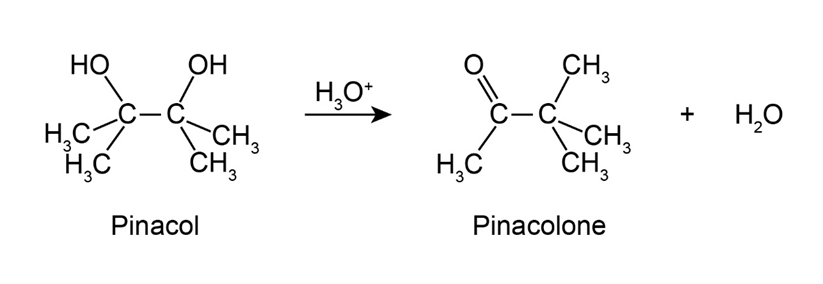
2,3-Dimethyl-2,3-butanediol has the common name pinacol. On heating with aqueous acid, pinacol rearranges to pinacolone, 3,3-dimethyl-2-butanone. Suggest a mechanism for this reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.