Chapter 17: Q15P (page 552)
What products would you expect from oxidation of the following compounds with in aqueous acid? With the Dess-Martin periodinane?
- 1-Hexanol
- 2-Hexanol
- Hexanal
Short Answer
a.
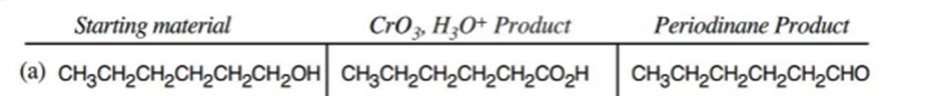
b.
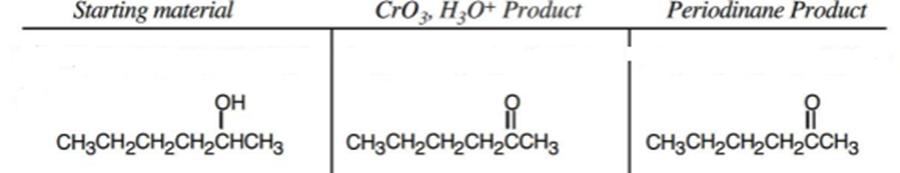
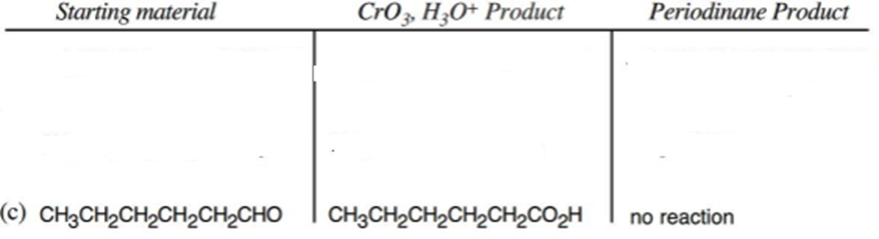
c.
Learning Materials
Features
Discover
Chapter 17: Q15P (page 552)
What products would you expect from oxidation of the following compounds with in aqueous acid? With the Dess-Martin periodinane?
a.
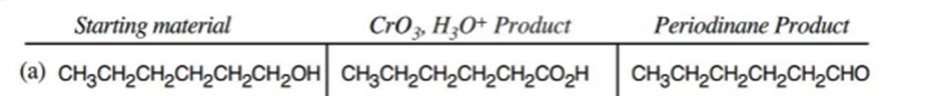
b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
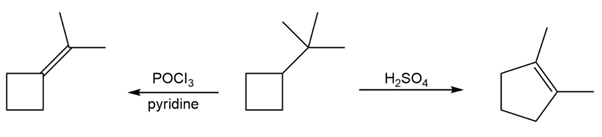
Question:When the alcohol below is treated with and pyridine, the expected elimination product is formed. However, when the same alcohol is treated with , the elimination product is 1,2-dimethylcyclopentene. Propose a mechanism for each pathway to account for these differences.
Compound A, , is one of the basic building blocks of nature. All steriods and many other naturally occurring compounds are built from compound A. Spectroscopic analysis of A yields the following information:
IR : ;
: 1.63 (3H, singlet); 1.70 (3H, singlet); 3.83 (1H, broad singlet); 4.15 (2H, doublet, J=7 Hz); 5.70 (1H, triplet, J = 7 Hz)
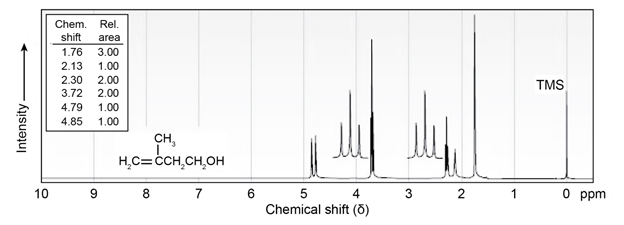
The NMR spectrum shown is that of 3-methyl-3-buten-1-ol. Assign
all the observed resonance peaks to specific protons, and account for
the splitting patterns.
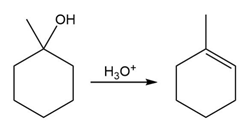
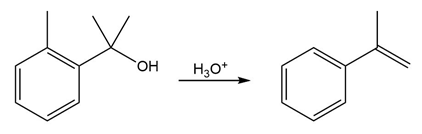
Question:The conversion of 3° alcohols into alkenes under acidic conditions involves two cationic intermediates. For each reaction, draw the complete mechanism using curved arrows.
a)
b)
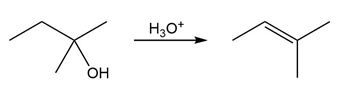
c)
The trimethylsilyl (TMS) protecting group is one of several silicon protecting groups for alcohols. For each reaction, draw the mechanism for the protection of (R)-3-bromo-1-butanol with the following silyl chlorides, using triethylamine as the base:
(a) tert-butyldimethylsilyl chloride (TBS-Cl)
(b) triisopropylsilyl chloride (TIPS-Cl)
(c) triethylsilyl chloride (TES-Cl)
What do you think about this solution?
We value your feedback to improve our textbook solutions.