Chapter 17: Q17-61E (page 567)
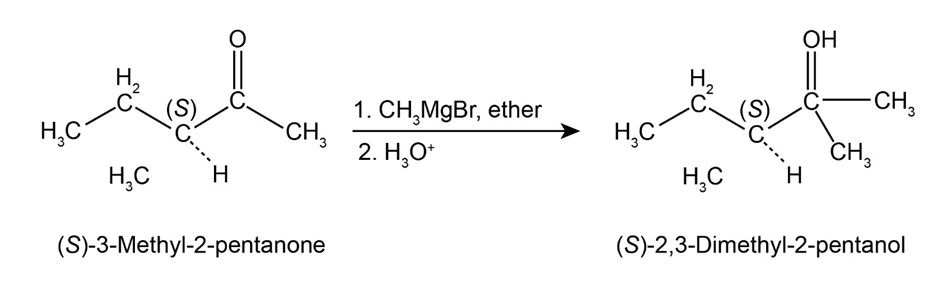
Question:Reaction of (S)-3-methyl-2-pentanone with methyl magnesium bromide followed by acidification yields 2,3-dimethyl-2-pentanol. What is the stereochemistry of the product? Is the product optically active?
Short Answer
(S)-3-methyl-2-pentanone treated with methylmagnesium bromide in the presence of ether followed by the hydrolysis gives (S)-2,3-dimethyl-2-pentanol.
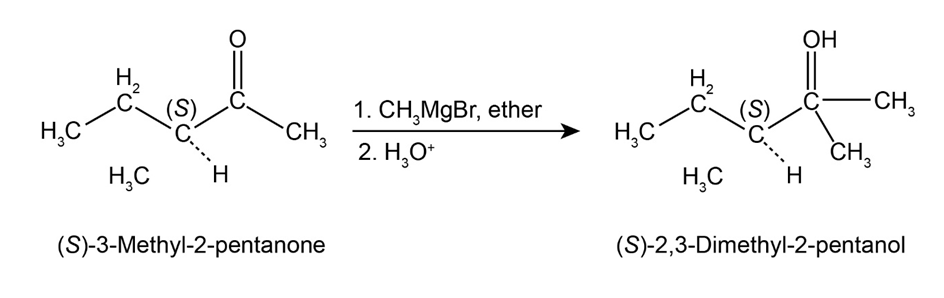
Here, the stereochemistry of the product does not change because the addition of Grignard reagent to the carbonyl compound does not produce a new chirality centre so there is no change at the chiral centre, which is already present. Hence, the product, which is formed, is an optically active compound.