Chapter 17: Q43E (page 567)
What carbonyl compounds would you reduce to prepare the followingalcohols? List all possibilities.
(a)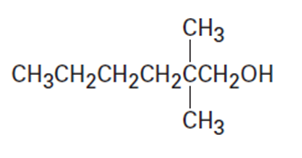
(b)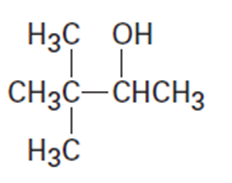
(c)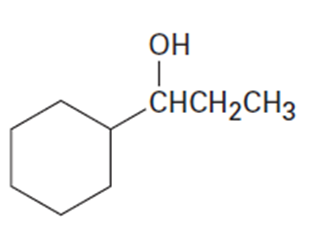
Short Answer
(a)
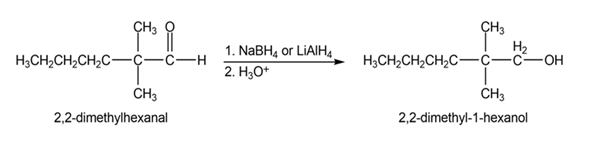
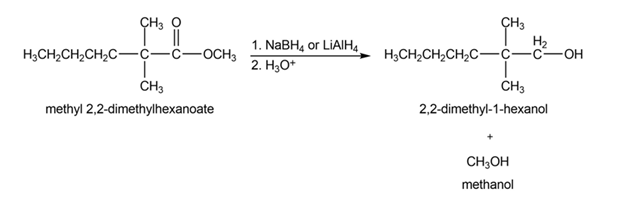
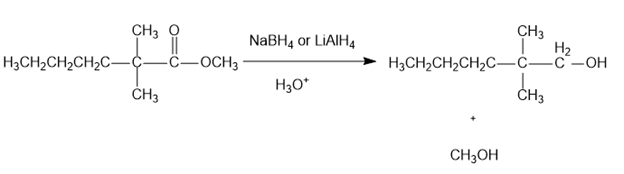
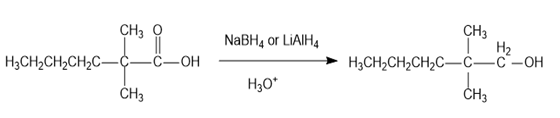
(b)
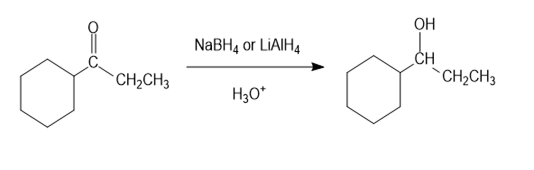
(c)
Learning Materials
Features
Discover
Chapter 17: Q43E (page 567)
What carbonyl compounds would you reduce to prepare the followingalcohols? List all possibilities.
(a)
(b)
(c)
(a)




(b)

(c)
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw and name the eight isomeric alcohols with the formula C5H12O
Question: What carbonyl compounds give the following alcohols on reduction with ? Show all possibilities.
(a)
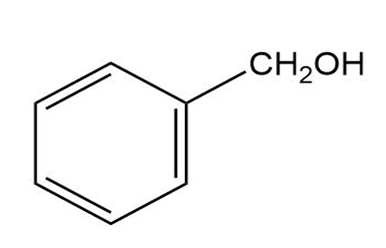
(b)
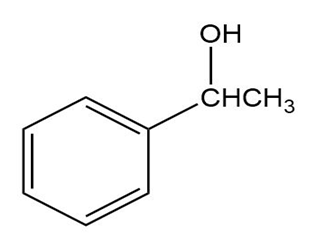
(c)
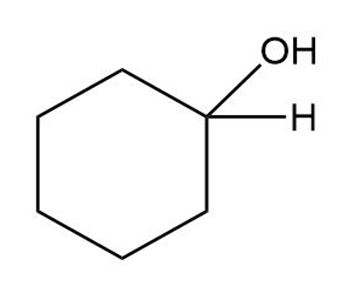
(d)
Question:The trimethylsilyl (TMS) protecting group is one of several silicon protecting groups for alcohols. For each reaction, draw the mechanism for the protection of (R)-3-bromo-1-butanol with the following silyl chlorides, using triethylamine as the base:
(a) tert-butyldimethylsilyl chloride (TBS-Cl)
(b) triisopropylsilyl chloride (TIPS-Cl)
(c) triethylsilyl chloride (TES-Cl)
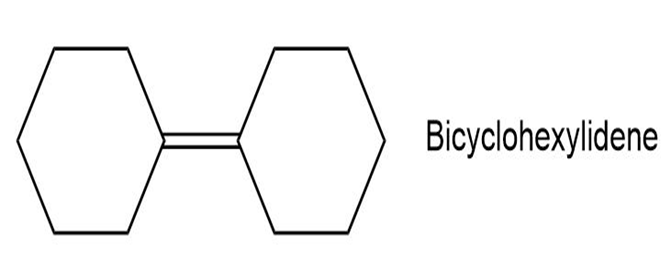
Propose a synthesis of bicyclohexylidene, starting from cyclohexanone as the only source of carbon.
Compound A, , undergoes reaction with dilute at to yield a mixture of two alkenes, . The major alkene product, B, gives only cyclopentanone after ozone treatment followed by reduction with zinc in acetic acid. Write the reactions involved, and identify A and B.
What do you think about this solution?
We value your feedback to improve our textbook solutions.