Chapter 17: Q17-75E (page 567)
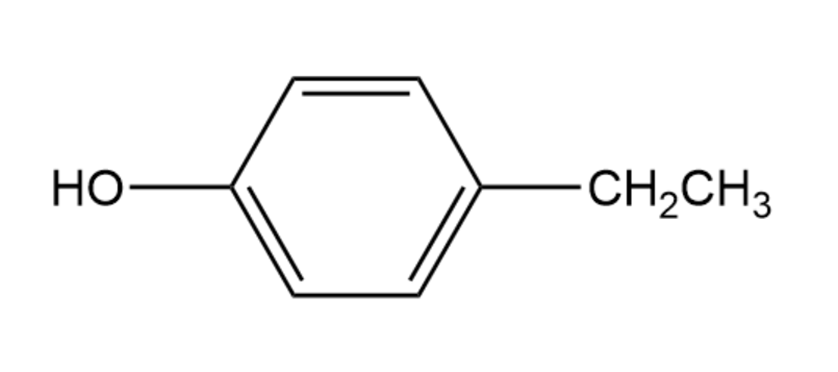
Compound A, , has the IR and NMR spectra shown. Proposea structure consistent with the observed spectra, and label each peak inthe NMR spectrum. Note that the absorption at disappears when is added.
Short Answer
Learning Materials
Features
Discover
Chapter 17: Q17-75E (page 567)
Compound A, , has the IR and NMR spectra shown. Proposea structure consistent with the observed spectra, and label each peak inthe NMR spectrum. Note that the absorption at disappears when is added.


All the tools & learning materials you need for study success - in one app.
Get started for free
Draw and name the eight isomeric alcohols with formula
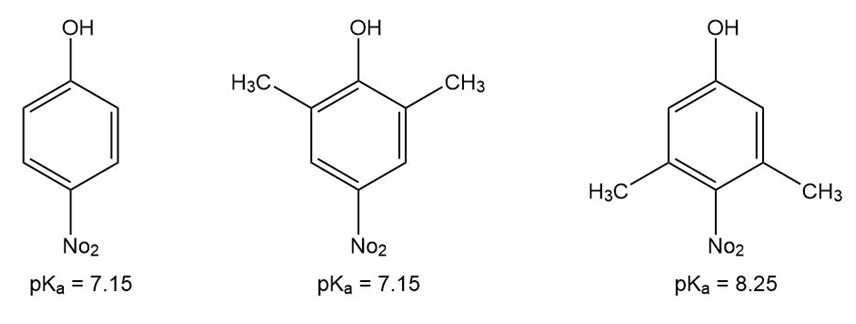
p-Nitrophenol and 2,6-dimethyl-4-nitrophenol both have pKa 5 7.15, but 3,5-dimethyl-4-nitrophenol has pKa 5 8.25. Why is 3,5-dimethyl-4- nitrophenol so much less acidic?
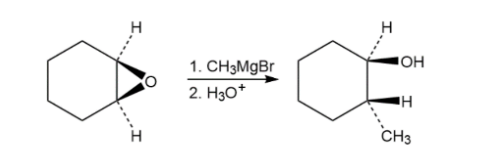
Epoxides react with Grignard reagents to yield alcohols. Propose a mechanism.
Question: Use a Grignard reaction to prepare the following alcohols.
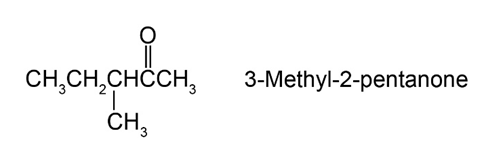
Question:Reaction of (S)-3-methyl-2-pentanone with methyl magnesium bromide followed by acidification yields 2,3-dimethyl-2-pentanol. What is the stereochemistry of the product? Is the product optically active?
What do you think about this solution?
We value your feedback to improve our textbook solutions.