Chapter 17: Q27E (page 567)
Epoxides react with Grignard reagents to yield alcohols. Propose a mechanism.
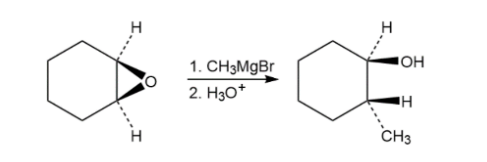
Short Answer
The mechanism followed as:
Learning Materials
Features
Discover
Chapter 17: Q27E (page 567)
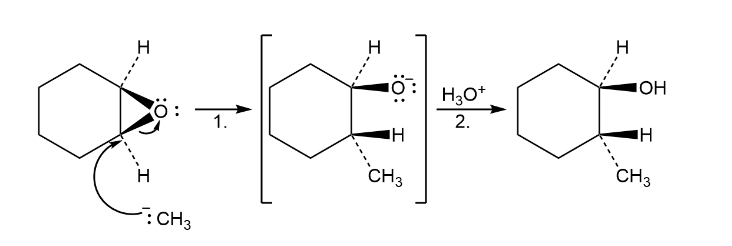
Epoxides react with Grignard reagents to yield alcohols. Propose a mechanism.

The mechanism followed as:

All the tools & learning materials you need for study success - in one app.
Get started for free
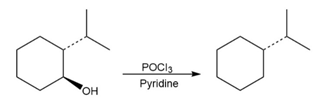
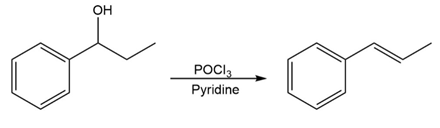
For each reaction, write the mechanism using curved arrows for the conversion of the alcohol into the corresponding alkene with POCl3. In each case, explain the regiochemistry of the elimination.
(a)
(b)
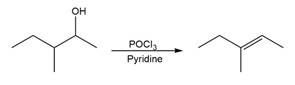
(c)
Draw and name the eight isomeric alcohols with formula
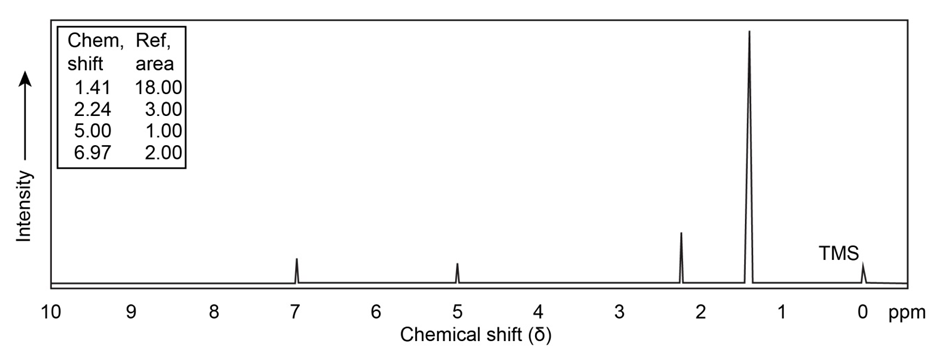
Propose a structure for a compound that has the followingNMR spectrum. The peak marked by an asterisk disappears whenis added to the sample.
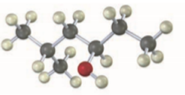
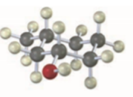
Give IUPAC names for the following compounds:
(a)
(b)
(c)
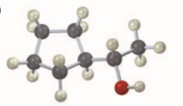
(d)
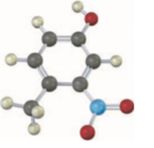
p-Nitrobenzyl alcohol is more acidic than benzyl alcohol, but p-methoxy-benzyl alcohol is less acidic. Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.