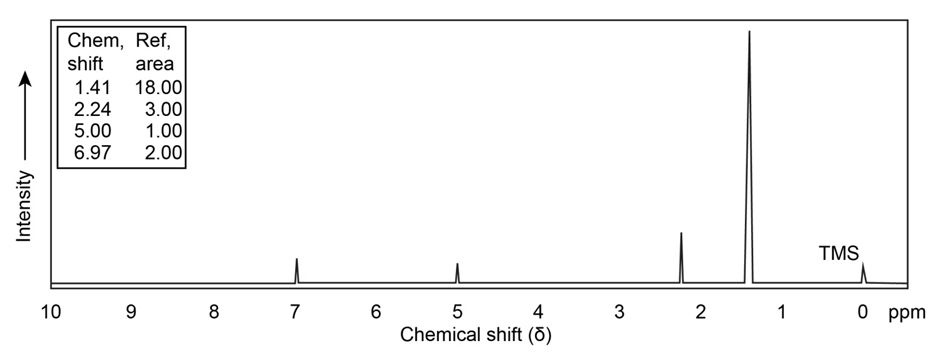
Chapter 17: Q17-56E (page 567)
Propose a structure for a compound that has the followingNMR spectrum. The peak marked by an asterisk disappears whenis added to the sample.
Short Answer
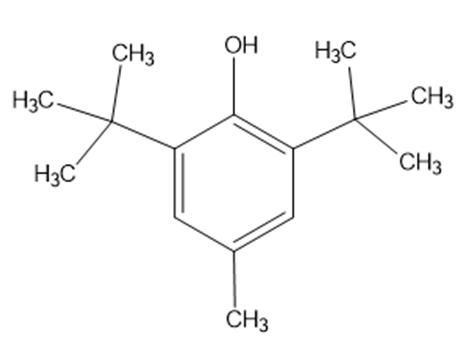
Butylated hydroxyl toluene(BHT)
Learning Materials
Features
Discover
Chapter 17: Q17-56E (page 567)
Propose a structure for a compound that has the followingNMR spectrum. The peak marked by an asterisk disappears whenis added to the sample.


Butylated hydroxyl toluene(BHT)
All the tools & learning materials you need for study success - in one app.
Get started for free
What product (s) would you expect from dehydration of the following alcohols with in pyridine? Indicate the major product in each case.
(a)
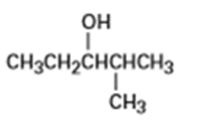
(b)
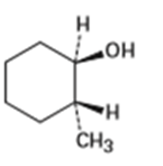
(C)
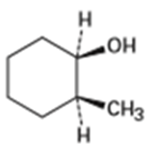
(d)
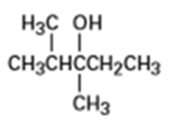
(e)
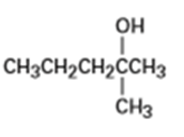
The trimethylsilyl (TMS) protecting group is one of several silicon protecting groups for alcohols. For each reaction, draw the mechanism for the protection of (R)-3-bromo-1-butanol with the following silyl chlorides, using triethylamine as the base:
(a) tert-butyldimethylsilyl chloride (TBS-Cl)
(b) triisopropylsilyl chloride (TIPS-Cl)
(c) triethylsilyl chloride (TES-Cl)
What products would you obtain from reaction of 1-pentanol with thefollowing reagents?
(a)
(b)
(c)
(d) Dess- Martin periodinane
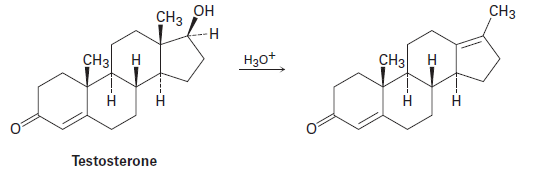
Testosterone is one of the most important male steroid hormones. When testosterone is dehydrated by treatment with acid, rearrangement occurs to yield the product shown. Propose a mechanism to account for this reaction.
Question:Identify the type of substitution mechanism ,involved in the conversion of the alcohol shown into the corresponding alkyl halide.
a.
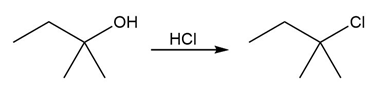
b)
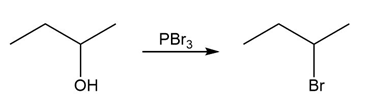
c)
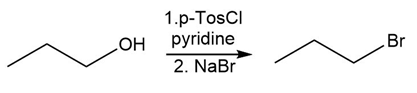
What do you think about this solution?
We value your feedback to improve our textbook solutions.