Chapter 17: 10 P (page 543)
Question: Use a Grignard reaction to prepare the following alcohols.
- 2-Methyl-2-propanol
- 1-Methylcyclohexanol
- 3-Methyl-3-pentanol
- 2-Phenyl-2-butanol
- Benzyl alcohol
- 4-Methyl-1-pentanol
Short Answer
(A)
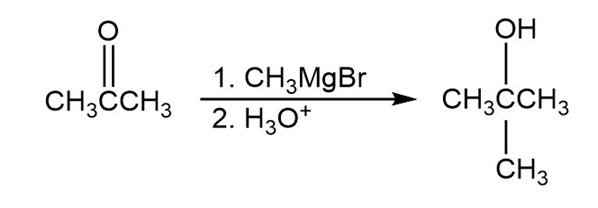
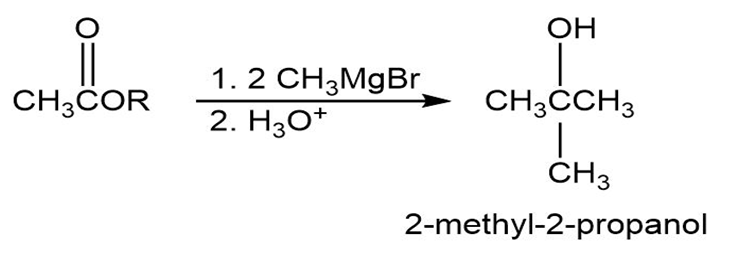
(B)
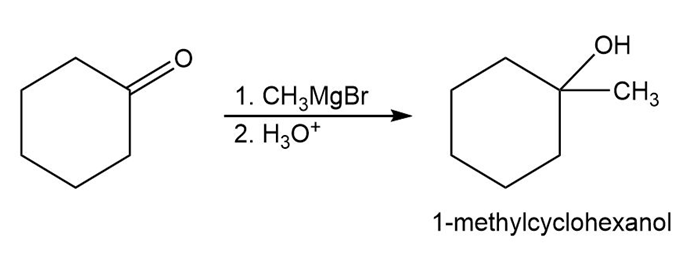
(C)
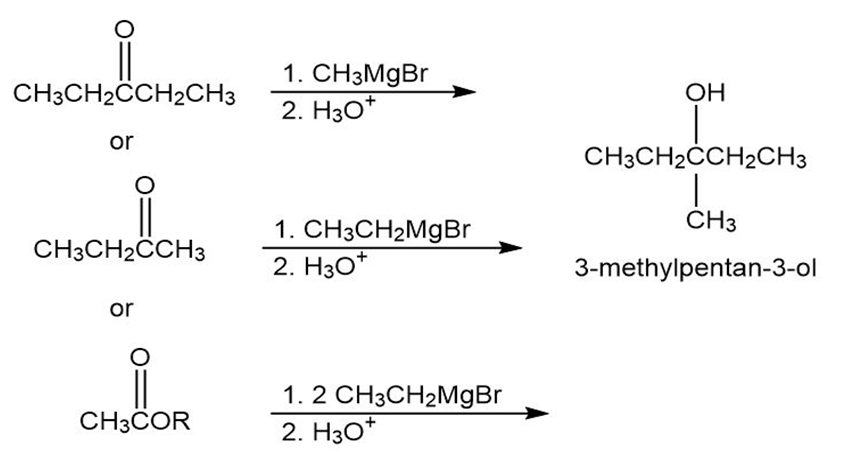
(D)
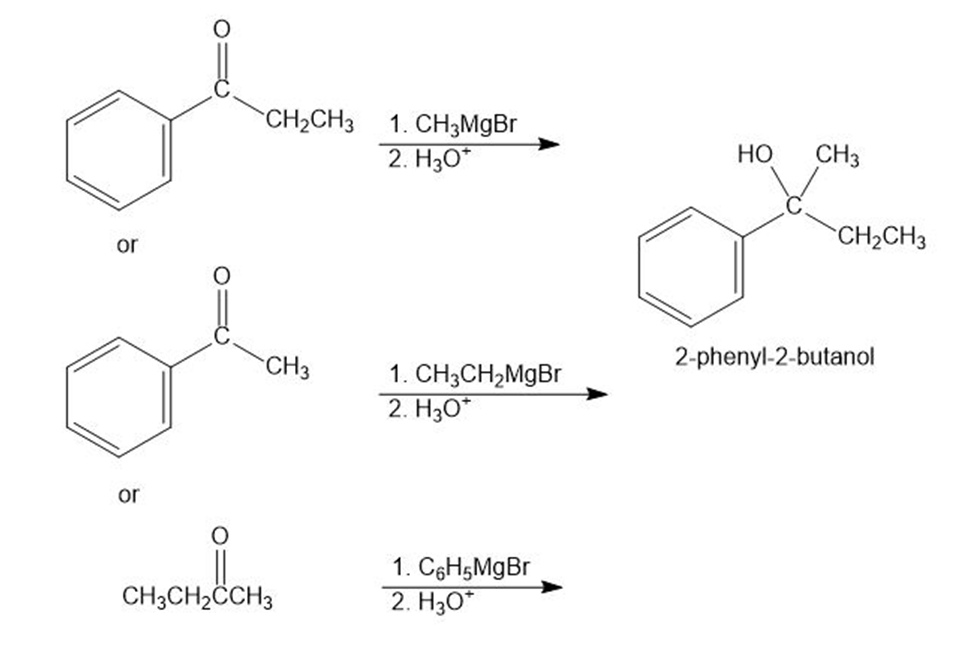
(E)
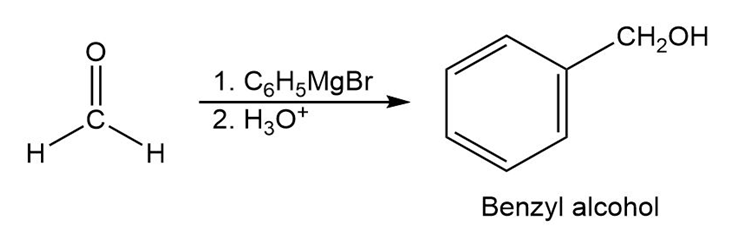
(F)
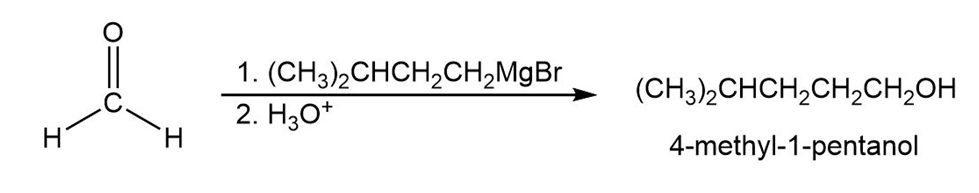
Learning Materials
Features
Discover
Chapter 17: 10 P (page 543)
Question: Use a Grignard reaction to prepare the following alcohols.
(A)


(B)

(C)

(D)

(E)

(F)

All the tools & learning materials you need for study success - in one app.
Get started for free
Dehydration of trans-2-methylcyclopentanol with POCl3 in pyridine yields predominantly 3-methylcyclopentene. Is the stereochemistry of this dehydration syn or anti? Can you suggest a reason for formation of the observed product? (Make molecular models!)
What products would you expect from oxidation of the following compounds with in aqueous acid? With the Dess-Martin periodinane?
Identify the type of substitution mechanism involved in the conversion of the alcohol shown into the corresponding alkyl halide.
(a)
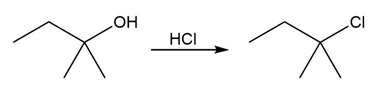
(b)
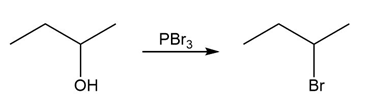
(c)
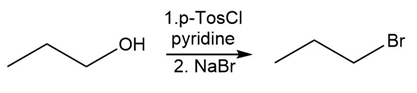
Question: How would you prepare the following compounds from
2-phenylethanol?
More than one step may be required.
(a) Styrene
(b) Phenylacetaldehyde
(c) Phenylacetic acid
(d) Benzoic acid
(e) Ethylbenzene
(f) Benzaldehyde
(g) 1-Phenylethanol
(h) 1-Bromo-2-phenylethane
What Grignard reagent and what carbonyl compound might you startwith to prepare the following alcohols?
(a)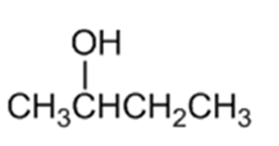
(b)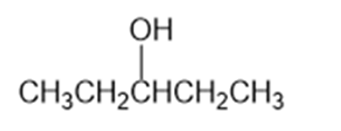
(c)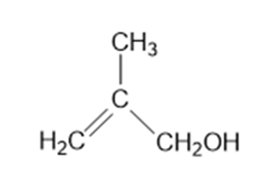
(d)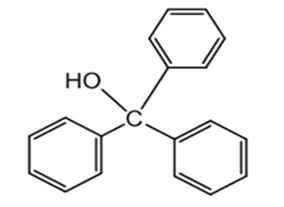
(e)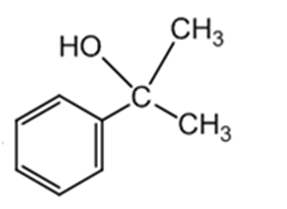
(f)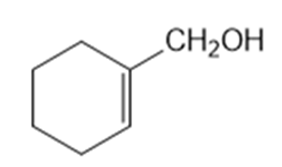
What do you think about this solution?
We value your feedback to improve our textbook solutions.