Chapter 17: Q12P (page 543)
How would you carry out the following transformation, a step used in the commercial synthesis of (S)-ibuprofen?
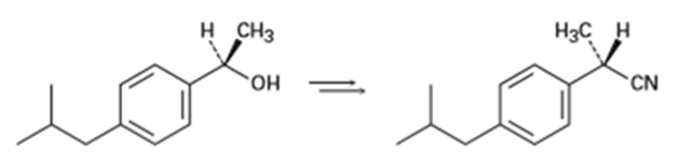
Short Answer
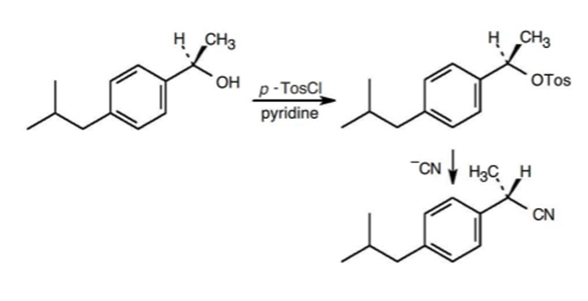
The transformation occurs in the commercial synthesis of (S)-ibuprofen is,
Learning Materials
Features
Discover
Chapter 17: Q12P (page 543)
How would you carry out the following transformation, a step used in the commercial synthesis of (S)-ibuprofen?

The transformation occurs in the commercial synthesis of (S)-ibuprofen is,

All the tools & learning materials you need for study success - in one app.
Get started for free
What products would you obtain from reaction of 1-pentanol with the
following reagents?
(a)
(b)
(c) , ,
(d) Dess–Martin periodinane
Draw structures corresponding to the following IUPAC names:
Question: Use a Grignard reaction to prepare the following alcohols.
Named bombykol, the sex pheromone secreted by the female silkworm
moth has the formula C16H28O and the systematic name (10E,12Z)-
10,12-hexadecadien-1-ol. Draw bombykol, showing the correct geometry
for the two double bonds.
Show the products obtained from addition of methylmagnesium bromide to the following compounds:
What do you think about this solution?
We value your feedback to improve our textbook solutions.