Chapter 17: Q23E (page 567)
Predict the product from reaction of the following substance with:
- ; then
- ; then
3. ; then
Short Answer
a.
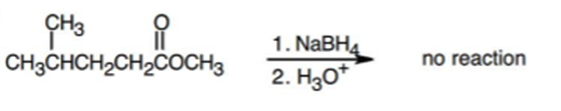
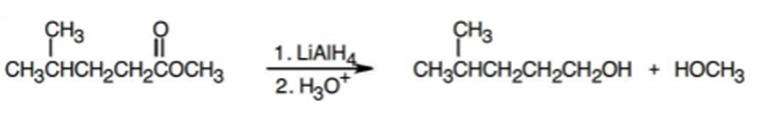
b.
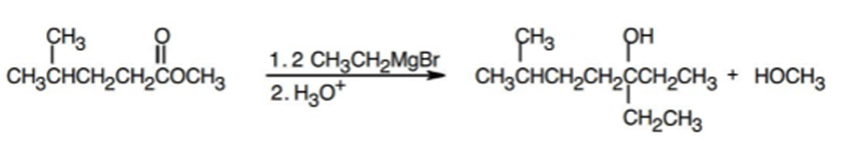
c.
Learning Materials
Features
Discover
Chapter 17: Q23E (page 567)
Predict the product from reaction of the following substance with:
3. ; then

a.
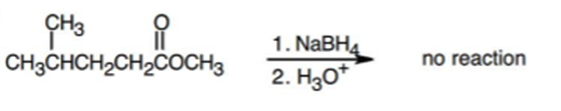
b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
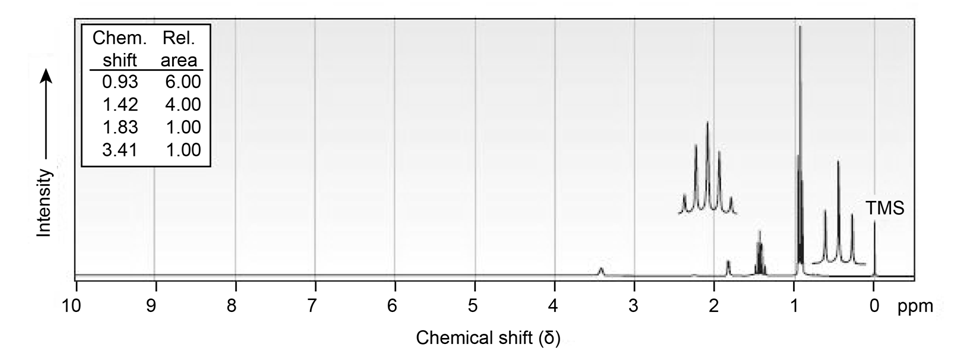
Propose structures for alcohols that have the following NMR spectra:
(a)
(b)

Question: Evidence for the intermediate carbocations in the acid-catalyzed dehydration of alcohols comes from the observation that rearrangements sometimes occur. Propose a mechanism to account for the formation of 2,3-dimethyl-2-butene from 3,3-dimethyl-2-butanol.

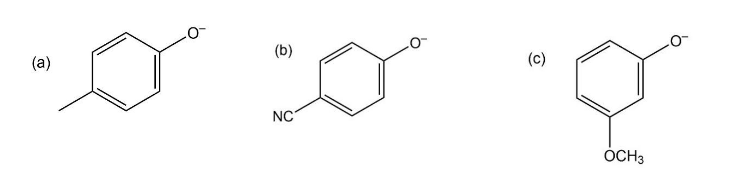
Phenols generally have lower pKa’s than aliphatic alcohols because of resonance stabilization with the aromatic ring. Draw all of the resonance contributors for the phenolate ions below. Make note of how the substituents either stabilize or destabilize the system.
As a rule, axial alcohols oxidize somewhat faster than equatorial alcohols. Which would you expect to oxidize faster, cis-4-tert-butylcyclohexanol or trans-4-tert-butylcyclohexanol? Draw the more stable chair conformation of each molecule.
Draw structures corresponding to the following IUPAC names:
What do you think about this solution?
We value your feedback to improve our textbook solutions.