Chapter 23: Substitution Reactions of Carbonyl Compounds at the α Carbon
Q56.
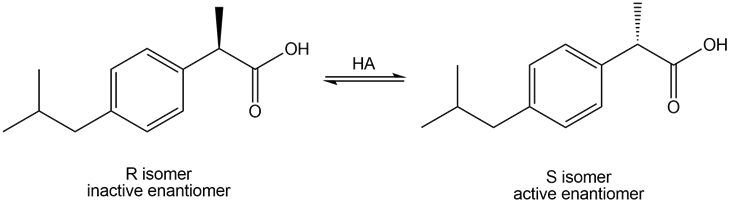
Question: Although ibuprofen is sold as a racemic mixture, only the S enantiomer acts as an analgesic. In the body, however, some of the R enantiomer is converted to the S isomer by tautomerization to an enol and then protonation to regenerate the carbonyl compound. Write a stepwise mechanism for this isomerization.
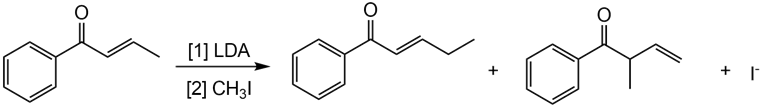
Q57.
Question: Draw a stepwise mechanism showing how two alkylation products are formed in the following reaction.
Q58.
Question: Draw a stepwise mechanism for the following reaction.
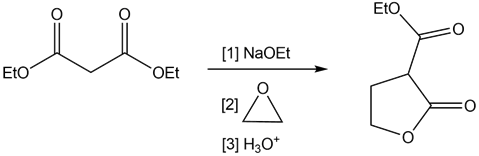
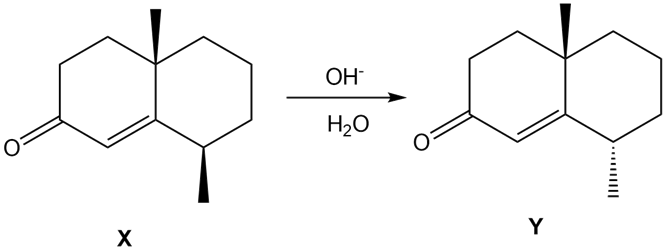
Q59.
Question: Treatment of α, β-unsaturated carbonyl compound X with base forms the diastereomer Y. Write a stepwise mechanism for this reaction. Explain why one stereogenic center changes configuration but the other does not.
Q6.
Question: Rank the protons in the labeled CH2groups in order of increasing acidity, and explain why you chose this order.
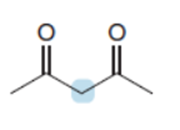
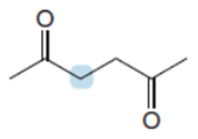
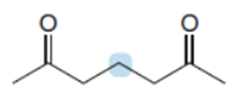
Q60.
Question: Draw stepwise mechanisms illustrating how each product is formed.
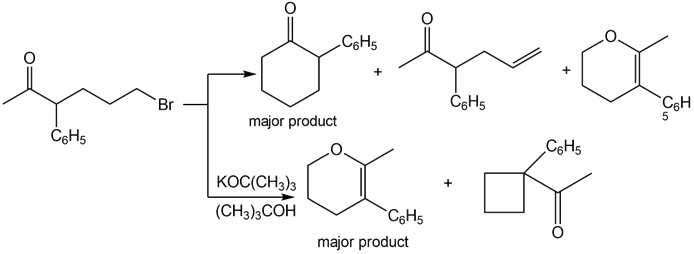
Q61.
Question: A key step in the synthesis of β-vetivone, a major constituent of vetiver, a perennial grass found in tropical and subtropical regions of the world, involved the reaction of compound A and dihalide B with two equivalents of LDA to form C. Draw a stepwise mechanism for this reaction. β-Vetivone contains a spiro ring system—that is, two rings that share a single carbon atom.
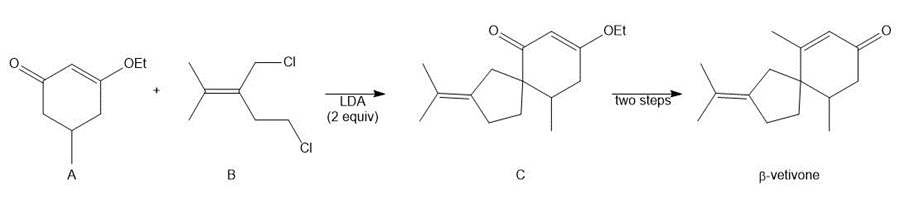
Q62.
Question:
(a) Draw two different haloketones that can form A by an intramolecular alkylation reaction.
(b) How can A be synthesized by an acetoacetic ester synthesis?
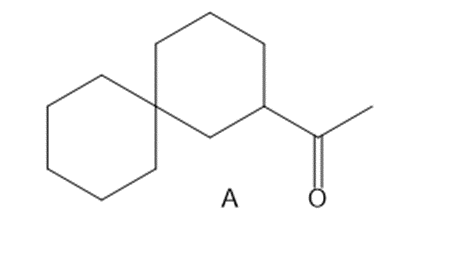
Q63.
Question: Synthesize each compound from cyclohexanone and organic halides having 4 C’s. You may use any other inorganic reagents.
a.
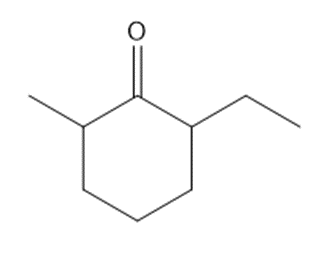
b.
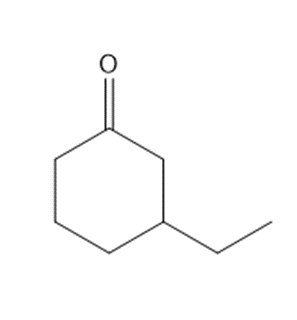
c.
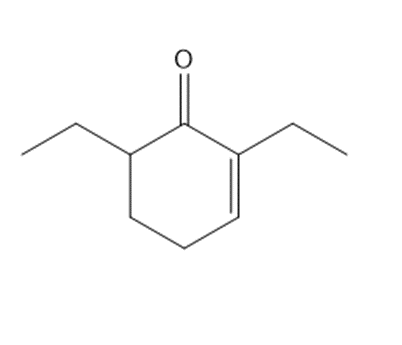
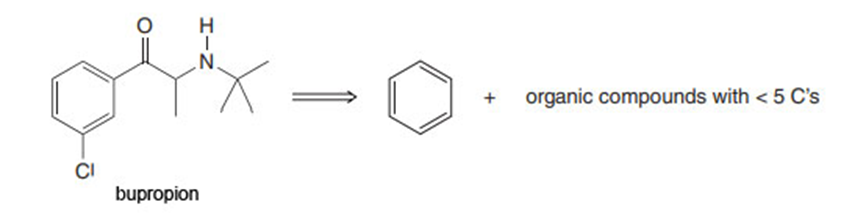
Q64.
Question:Bupropion, sold under the trade name of Zyban, is an antidepressant that was approved to aid smoking cessation in 1997. Devise a synthesis of bupropion from benzene, organic compounds that have fewer than five carbons, and any required inorganic reagents.