Chapter 23: Q58. (page 924)
Question: Draw a stepwise mechanism for the following reaction.
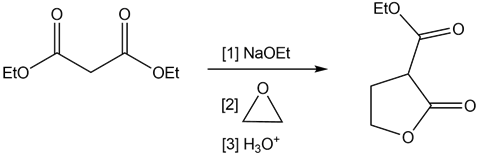
Short Answer
Answer
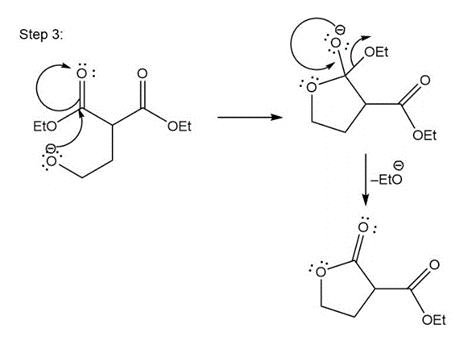
Stepwise mechanism


Learning Materials
Features
Discover
Chapter 23: Q58. (page 924)
Question: Draw a stepwise mechanism for the following reaction.
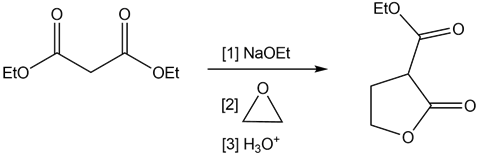
Answer
Stepwise mechanism



All the tools & learning materials you need for study success - in one app.
Get started for free
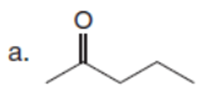
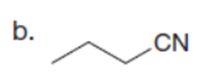
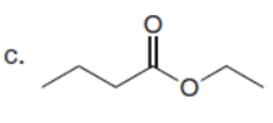
Question:What alkyl halides are needed to prepare each ketone using the acetoacetic ester synthesis?
Question: Draw a stepwise mechanism for the following reaction.
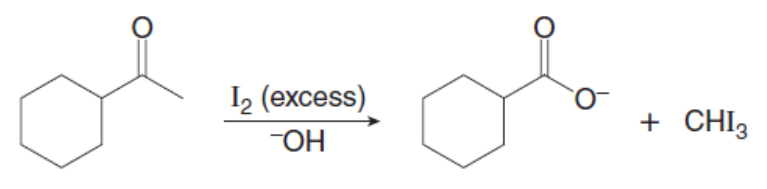
Question: Treatment of ethyl acetoacetate with NaOEt (2 equiv) and BrCH2CH2Brforms compound X. This reaction is the first step in the synthesis of illudin-S, an antitumor substance isolated from thejack-o’-lantern, a poisonous, saffron-colored mushroom. What is the structure of X?
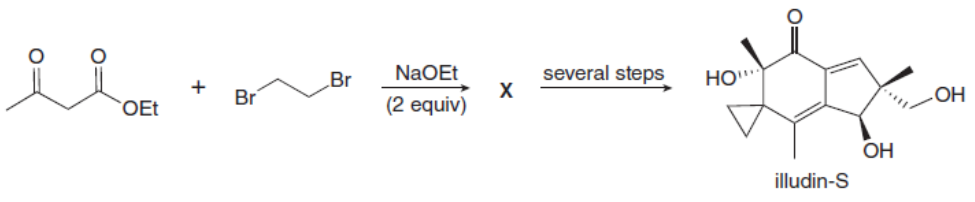
Question: Which C-Hbonds in the following molecules are acidic because the resulting conjugate base is resonance stabilized?

Question: A key step in the synthesis of the narcotic analgesic meperidine (trade name Demerol) is the conversion of phenyl acetonitrile to X. (a) What is the structure of X? (b) What reactions convert X to meperidine?

What do you think about this solution?
We value your feedback to improve our textbook solutions.