Chapter 26: Carbon-Carbon Bond-Forming Reactions in Organic Synthesis
Q 1.
Draw the product of each coupling reaction.
a.
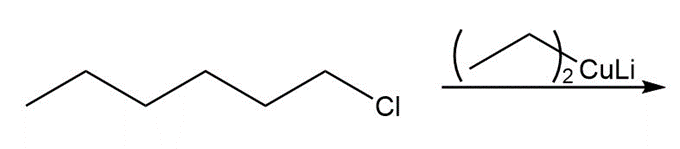
b.
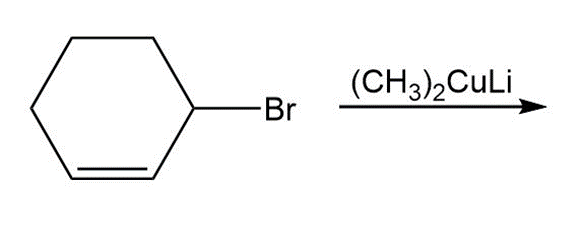
c.
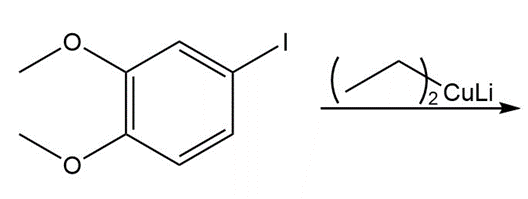
d.
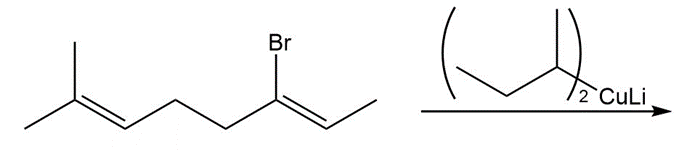
Q 10.
What reagents are needed to convert 2-methylpropene to each compound? More than one step may be required.
a.
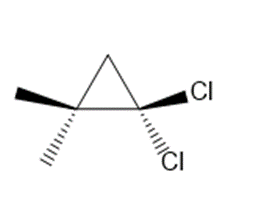
b.
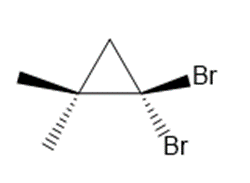
c.
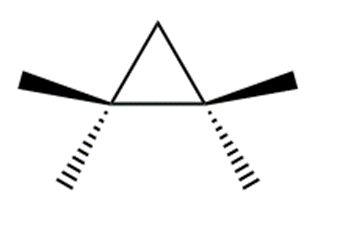
Q 11
What product is formed when each alkene is treated with and Zn(Cu)?
a.
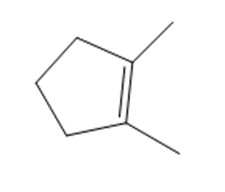
b.
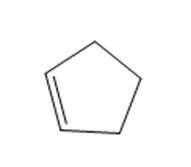
c.
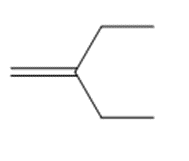
Q 12.
What stereoisomers are formed when trans-hex-3-ene is treated with and Zn(Cu)?
Q 13.
Draw the products formed when each alkene is treated with Grubbs catalyst.
a.
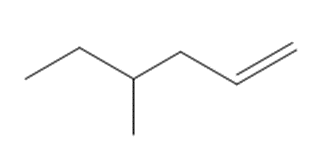
b.
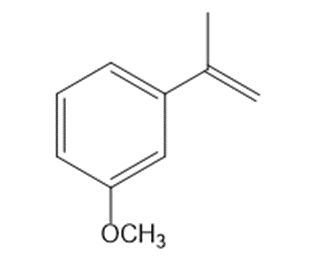
c.
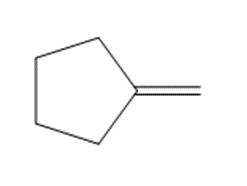
Q 14
What products are formed when cis-pent-2-ene undergoes metathesis? Use this reaction to explain why metathesis of a 1, 2-disubstituted alkene is generally not a practical method for alkene synthesis?
Q 15.
Draw the product formed from ring-closing metathesis of each compound.
a.
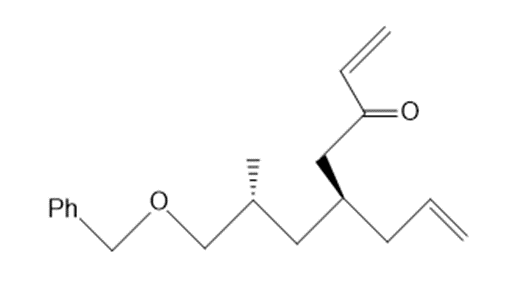
b.
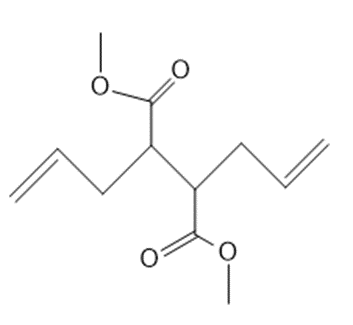
Q 16.
What product is formed by ring-closing metathesis of compound V, a key intermediate in the synthesis of ingenol, a natural product mentioned in the chapter opener?
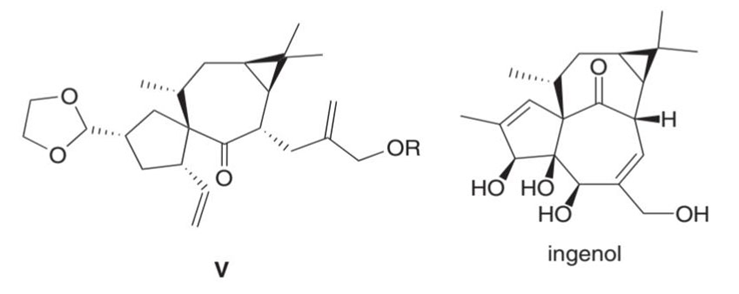
Q 17.
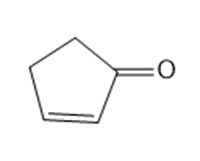
What starting material is needed to synthesize each compound by a ring-closing metathesis reaction?
a.
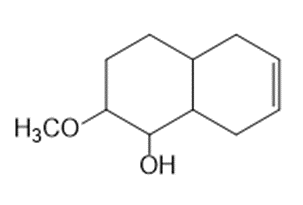
b.
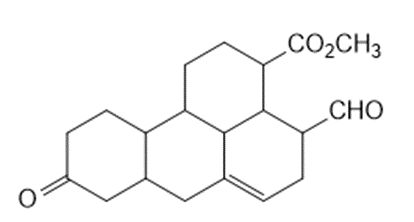
c.
Q 18.
In addition to organic halides, alkyl tosylates (R'OTs, Section 9.13) also react with organocuprates ( ) to form coupling products R-R'. When 2° alkyl tosylates are used as starting materials ( ), inversion of the configuration at a stereogenic center results. Keeping this in mind, draw the product formed when each compound is treated with .
a.
b.