Chapter 16: Conjugation, Resonance, and Dienes
PROBLEM 16.1
Question: Classify each diene as isolated or conjugated.
a.
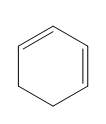
b.
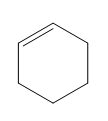
c.
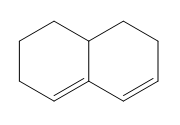
d.
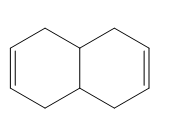
PROBLEM 16.10
Question. Draw the structure consistent with each description.
a. (2E,4E)-octa-2,4-diene in the s-trans conformation
b. (3E,5Z)-nona-3,5-diene in the s-cis conformation
c. (3Z,5Z)-4,5-dimethyldeca-3,5-diene. Draw both the s-cis and s-trans conformations.
PROBLEM 16.11
Question: Neuroprotectin D1 (NPD1) is synthesized in the body from highly unsaturated essential fatty acids. NPD1 is a potent natural anti-inflammatory agent.
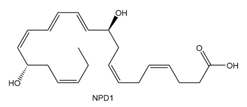
a. Label each double bond as conjugated or isolated.
b. Label each double bond as E or Z.
c. For each conjugated system, label the given conformation as s-cis or s-trans.
PROBLEM 16.12
Question: Using hybridization, predict how the bond length of the C-Cσ bond in should compare with the C-Cσ bond inand.
PROBLEM 16.13
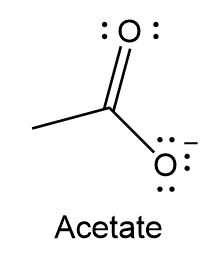
Question: Use resonance theory explains why the labeled C-O bond lengths are equal in the acetate ion.
PROBLEM 16.15
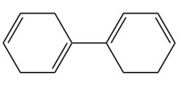
Question: Rank the following compounds in order of increasing stability.
a.
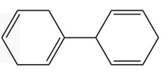
b.
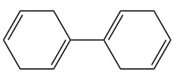
c.
PROBLEM 16.16
Question: Draw the products formed when each diene is treated with one equivalent of HCl.
a.
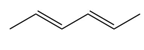
b.
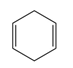
c.

d.

PROBLEM 16.17
Question: Draw a stepwise mechanism for the following reaction.
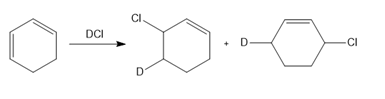
PROBLEM 16.18
Question: Label each product in the following reaction as a 1,2-product or 1,4-product, and decide which is the kinetic product and which is the thermodynamic product.
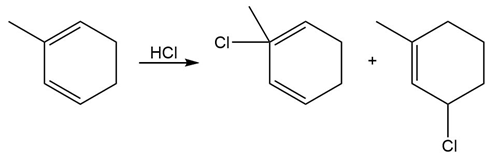
PROBLEM 16.19
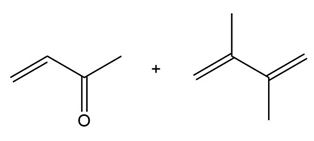
Question: Draw the product formed when each diene and dienophile react in a Diels–Alder reaction.
a.
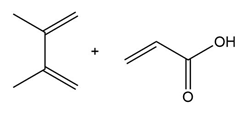
b.
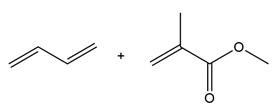
c.