Chapter 12: Oxidation and Reduction
PROBLEM 12.1
Question: Classify each reaction as oxidation, reduction, or neither
a.
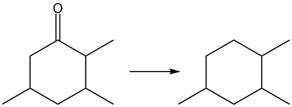
b.
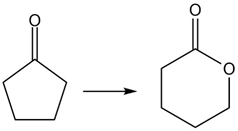
c.
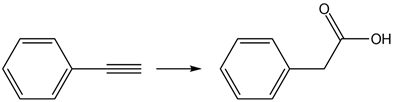
d.
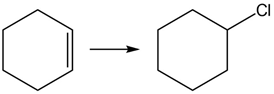
PROBLEM 12.10
Question: What is the structure of cis-jasmone, a natural product isolated from jasmine flowers, formed by the treatment of alkyne A with H2in the presence of the Lindlar catalyst?
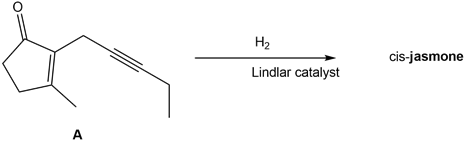
PROBLEM 12.11
Question: a) Draw the structure of a compound of molecular formula C6H10 that reacts with H2 in the presence of Pd-C but does not react with H2in the presence of Lindlar catalyst. (b) Draw the structure of a compound of molecular formula C6H10 that reacts with H2 when either catalyst is present
PROBLEM 12.12
Question: What product is formed when is treated with each reagent: (a) H2 (excess), Pd-C; ; (b) H2(1 equiv), Lindlar catalyst; (c) H2(excess), Lindlar catalyst; (d) Na,NH3 ?
PROBLEM 12.13
Question: A chiral alkyne A with molecular formula C6H10 is reduced with and Lindlar catalyst to B having the R configuration at its stereogeniccentre. What are the structures of A and B?
PROBLEM 12.14
Question: Draw the products of each reaction
a.
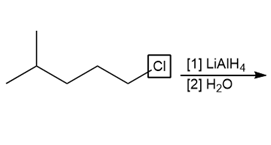
b.
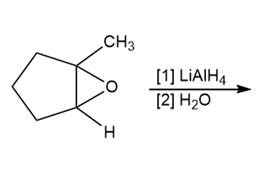
PROBLEM 12.15
Question:What epoxide is formed when each alkene is treated with mCPBA?
a.
b.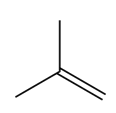
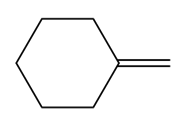
c.
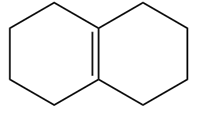
PROBLEM 12.16
Question: Draw all stereoisomers formed when each alkene is treated with mCPBA.
a.
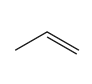
b.
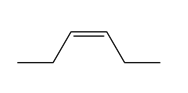
c.
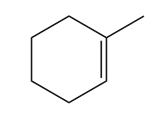
PROBLEM 12.17
Question: Draw the products formed when both cis- and trans-but-2-ene are treated with a peroxyacid followed by – OH (in H2O ). Explain how these reactions illustrate that anti-dihydroxylation is stereospecific.
PROBLEM 12.18
Question: Draw the products formed when both cis- and trans-but-2-ene are treated with OsO4, followed by hydrolysis with NaHSO3+H2O. Explain how these reactions illustrate that syndihydroxylation is stereospecific.
