Chapter 12: Oxidation and Reduction
Q.61.
Question: Devise a synthesis of muscalure, the sex pheromone of the common housefly, from acetylene and any other required reagents.
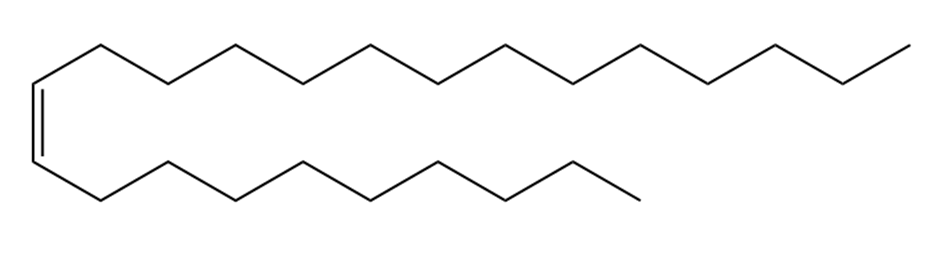
Muscalure
Q.62.
Question: It is sometimes necessary to isomerize a cis alkene to a trans alkene in a synthesis, a process that cannot be accomplished in a single step. Using the reactions that you have learned in Chapters 8–12, devise a stepwise method to convert cis-but-2-ene to trans-but-2-ene.
Q.63.
Question:Devise a synthesis of each compound from acetylene and any other required reagents.
Q.64.
Question:Devise a synthesis of compound A from the given starting materials. You may use any other inorganic reagents or organic alcohols. A was used to prepare aliskiren, a drug used to treat hypertension (see also Problem 5.7).
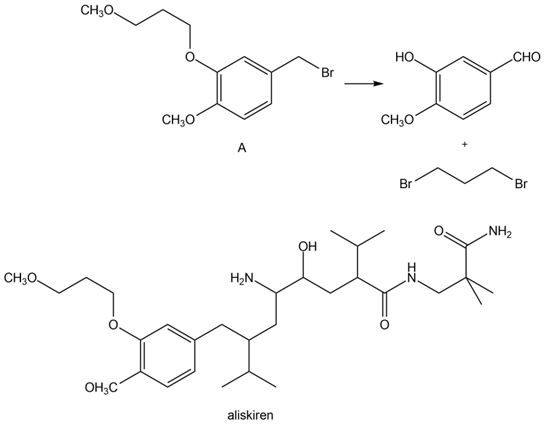
Q.65.
Question:Devise a synthesis of (E)-hex-2-ene from pent-1-ene and any needed organic compounds or inorganic reagents.
Q.66.
Question: Devise a synthesis of each compound from the indicated starting material, organic compounds containing one or two carbons, and any other required reagents.
Q.67.
Question:Devise a synthesis of 1-phenyl-5-methylhexane from acetylene, alkylhalides, and any required inorganic reagents.
Q.68.
Question:Devise a synthesis of (3R,4S)-3,4-dichlorohexane from acetylene and any needed organic compounds or inorganic reagents.
Q.69.
Question: Devise a synthesis of each compound from as the only organic starting material; that is, every carbon in the product must come from a molecule of ethanol. You may use any other needed inorganic reagents.
Q.70.
Question: The Birch reduction is a dissolving metal reaction that converts substituted benzenes to cyclohexa-1,4-dienes using Li and liquid ammonia in the presence of an alcohol. Draw a stepwise mechanism for the following Birch reduction.




