Chapter 12: Q.65. (page 493)
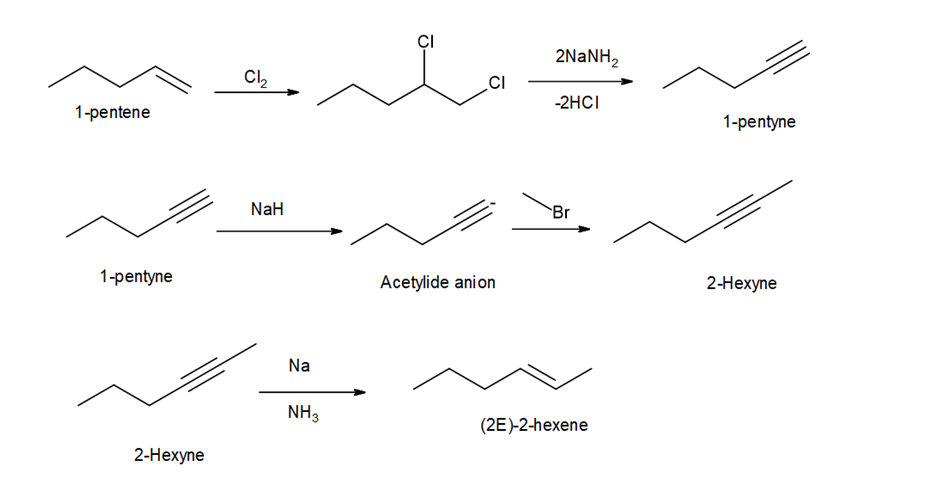
Question:Devise a synthesis of (E)-hex-2-ene from pent-1-ene and any needed organic compounds or inorganic reagents.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 12: Q.65. (page 493)
Question:Devise a synthesis of (E)-hex-2-ene from pent-1-ene and any needed organic compounds or inorganic reagents.
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
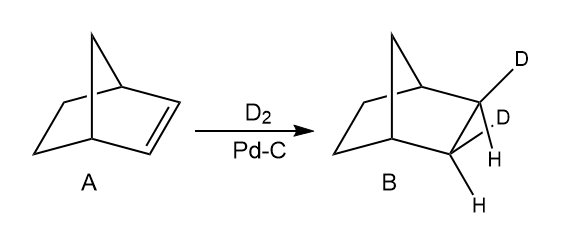
Question: Hydrogenation of alkene A with D2 in the presence of Pd-C affords a single product B. Keeping this result in mind, what compound is formed when A is treated with each reagent:
(a) mCPBA; (b) Br2 , H2O followed by base? Explain these results.
Question: What allylic alcohol and DET isomer are needed to make each chiral epoxide using a sharpless asymmetric epoxidation reaction?
Question: Draw the structure of two different epoxides that would yield 2-methylpentan-2-ol [ (CH3)2CH(OH)CH2CH2CH3] when reduced with LiAlH4 .
Question: Devise a synthesis of muscalure, the sex pheromone of the common housefly, from acetylene and any other required reagents.
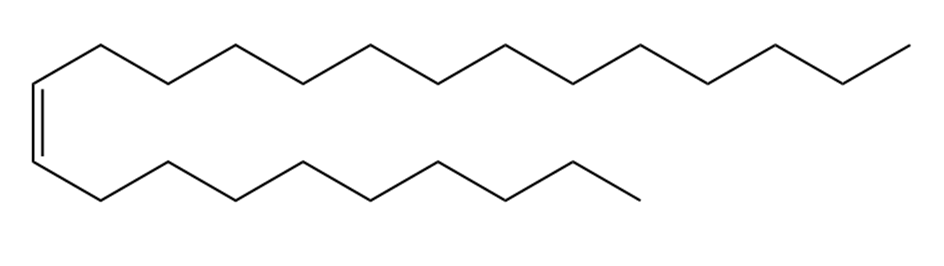
Muscalure
Question: Draw the products formed when both cis- and trans-but-2-ene are treated with a peroxyacid followed by – OH (in H2O ). Explain how these reactions illustrate that anti-dihydroxylation is stereospecific.
What do you think about this solution?
We value your feedback to improve our textbook solutions.