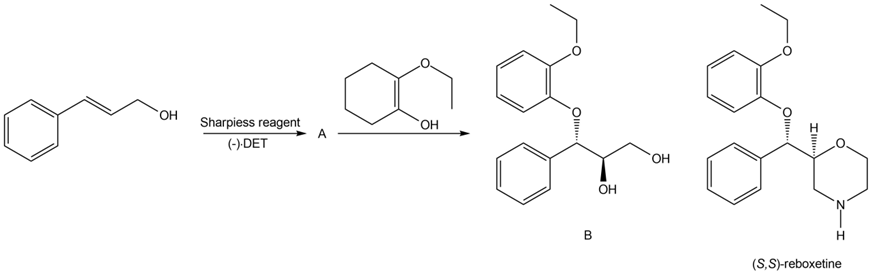
Chapter 12: PROBLEM 12.42 (page 489)
Question: Draw the structure of two different epoxides that would yield 2-methylpentan-2-ol [ (CH3)2CH(OH)CH2CH2CH3] when reduced with LiAlH4 .
Short Answer
Answer
 Epoxide 1- 2-methyl-2-propyl oxirane
Epoxide 1- 2-methyl-2-propyl oxirane
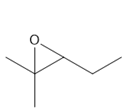
Epoxide 2- 3-ethyl-2,2-dimethyloxirane
 Epoxide 1- 2-methyl-2-propyl oxirane
Epoxide 1- 2-methyl-2-propyl oxirane Epoxide 2- 3-ethyl-2,2-dimethyloxirane
Epoxide 2- 3-ethyl-2,2-dimethyloxirane