Chapter 12: Q.68. (page 493)
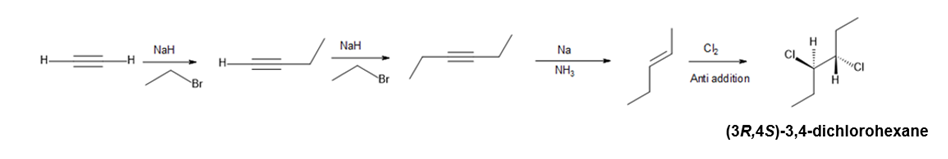
Question:Devise a synthesis of (3R,4S)-3,4-dichlorohexane from acetylene and any needed organic compounds or inorganic reagents.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 12: Q.68. (page 493)
Question:Devise a synthesis of (3R,4S)-3,4-dichlorohexane from acetylene and any needed organic compounds or inorganic reagents.
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Devise a synthesis of each compound from as the only organic starting material; that is, every carbon in the product must come from a molecule of ethanol. You may use any other needed inorganic reagents.
Question: What alkane is formed when each alkene is treated with H2 and a Pd catalyst?
a.
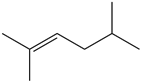
b.
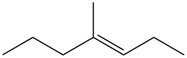
c.
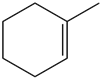
Question: What product is formed when is treated with each reagent: (a) H2 (excess), Pd-C; ; (b) H2(1 equiv), Lindlar catalyst; (c) H2(excess), Lindlar catalyst; (d) Na,NH3 ?
Question: Draw the products formed when each diene is treated with O3 followed by CH3SCH3.
a.
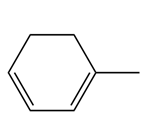
b.
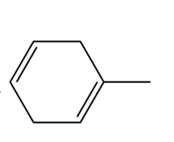
c.
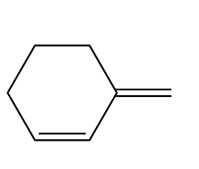
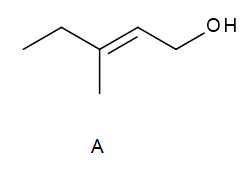
Question: Draw the organic products formed when allylic alcohol A is treated with each reagent.
a. H2 +Pd-c
b. mCPBA
c. PCC
d. CrO3 ,H2SO4, H2O
e.(CH3)3COOH , Ti [OCH(CH3)3]4 (+) -DET
f.(CH3)3COOH , Ti [OCH(CH3)3]4 (-) -DET
g. [1] PBr3,[2] LiAlH4 ,[3] H2O
h. HCrO4- –Amberlyst A-26 resin
What do you think about this solution?
We value your feedback to improve our textbook solutions.