Chapter 12: PROBLEM 12.2 (page 458)
Question: What alkane is formed when each alkene is treated with H2 and a Pd catalyst?
a.
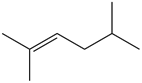
b.
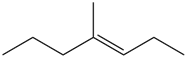
c.
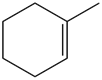
Short Answer
Answer
a.
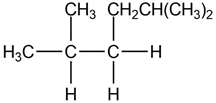
b.
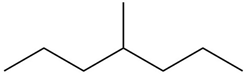
c.
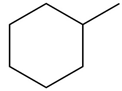
Learning Materials
Features
Discover
Chapter 12: PROBLEM 12.2 (page 458)
Question: What alkane is formed when each alkene is treated with H2 and a Pd catalyst?
a.

b.

c.

Answer
a.

b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
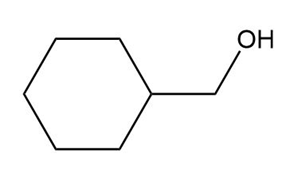
Question: What carbonyl compound is formed when each alcohol is treated with HCrO4––Amberlyst A-26 resin?
a.
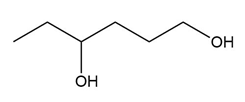
b.
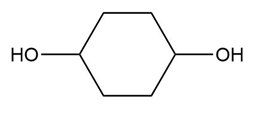
c.
Question: Stearidonic acid (C18H28O2 ) is an unsaturated fatty acid obtained from oils isolated from hemp and blackcurrant (see also Problem 10.11).
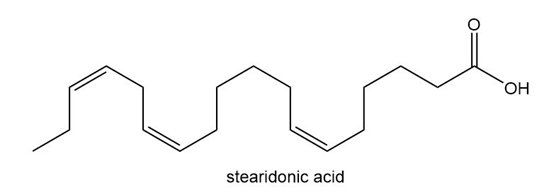
a. What fatty acid is formed when Stearidonic acid is hydrogenated with excess H2 and a Pd catalyst?
b. What fatty acids are formed when stearidonic acid is hydrogenated with one equivalent of H2 and a Pd catalyst?
c. Draw the structure of a possible product formed when stearidonic acid is hydrogenated with one equivalent of H2 and a Pd catalyst, and one double bond is isomerized to a trans isomer.
d. How do the melting points of the following fatty acids compare: stearidonic acid; one of the products formed in part (b); the product drawn in part (c)?
Question:Devise a synthesis of (3R,4S)-3,4-dichlorohexane from acetylene and any needed organic compounds or inorganic reagents.
Question: Sodium hypochlorite (NaOCl, the oxidant in household bleach) in aqueous CH3COOH is also touted as a “green” oxidizing agent. For example, oxidation of (CH3)2CHOH with NaOCl forms along with NaCl and H2O. (a) What advantages and/or disadvantages does this method have over oxidation with HCrO4––Amberlyst A-26 resin? (b) What advantages and/or disadvantages does this method have over oxidation with CrO3, H2SO4,H2O.
Question: Draw the products formed when both cis- and trans-but-2-ene are treated with a peroxyacid followed by – OH (in H2O ). Explain how these reactions illustrate that anti-dihydroxylation is stereospecific.
What do you think about this solution?
We value your feedback to improve our textbook solutions.