Chapter 12: PROBLEM 12.25 (page 480)
Question: What carbonyl compound is formed when each alcohol is treated with HCrO4––Amberlyst A-26 resin?
a.
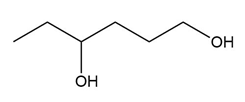
b.
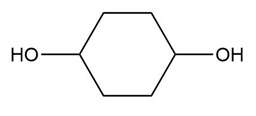
c.
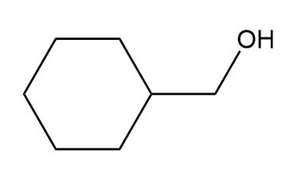
Short Answer
Answer
a.
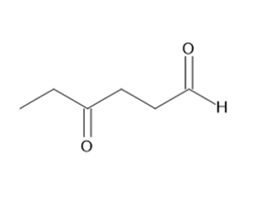
b.
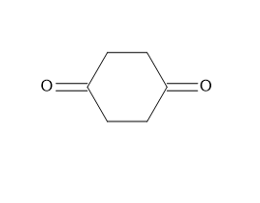
c.
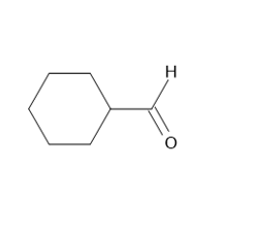
Learning Materials
Features
Discover
Chapter 12: PROBLEM 12.25 (page 480)
Question: What carbonyl compound is formed when each alcohol is treated with HCrO4––Amberlyst A-26 resin?
a.

b.

c.

Answer
a.

b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Oximene and myrcene, two hydrocarbons isolated from alfalfa that have the molecular formula , both yield 2,6-dimethyloctane when treated with and a Pd catalyst. Ozonolysis of oximene forms , , and . Ozonolysis of myrcene yields , (two equiv), and . Identify the structures of oximene and myrcene.
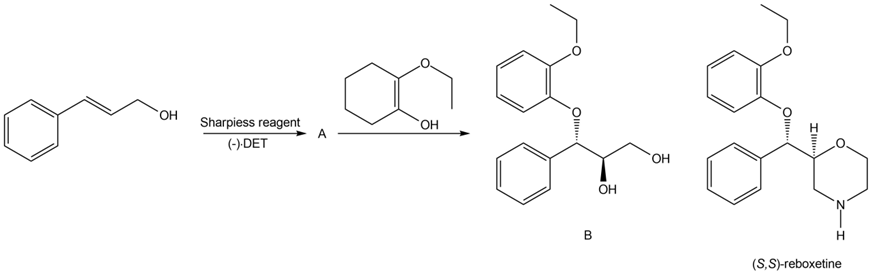
Question: Identify A in the following reaction sequence and draw a mechanism for the conversion of A to B. B has been converted to (S,S)-reboxetine, an antidepressant marketed outside the United States.
Question: Draw the organic products formed in each reaction.
a.
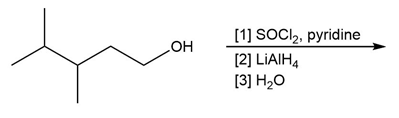
b.
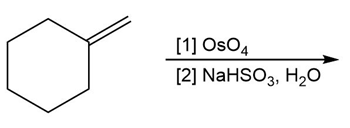
c.
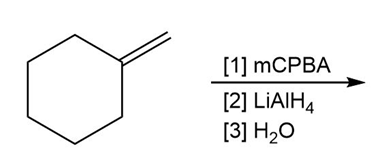
d.
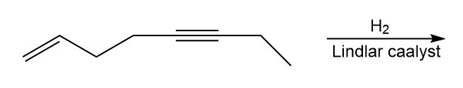
Question: What allylic alcohol and DET isomer are needed to make each chiral epoxide using a sharpless asymmetric epoxidation reaction?
Question: In the oxidation of cyclohexanols, it is generally true that sterically hindered alcohols react faster than unhindered alcohols. Which of the following alcohols should be oxidized more rapidly?
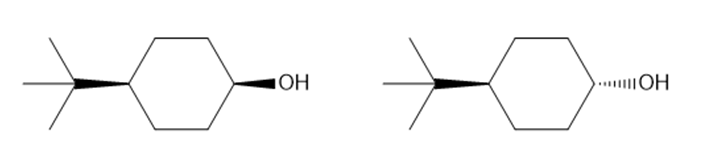
What do you think about this solution?
We value your feedback to improve our textbook solutions.