Chapter 12: Q.53. (page 491)
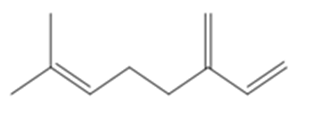
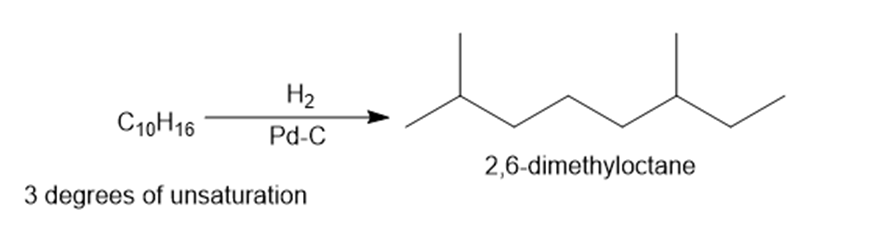
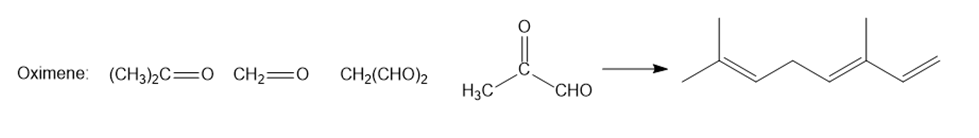
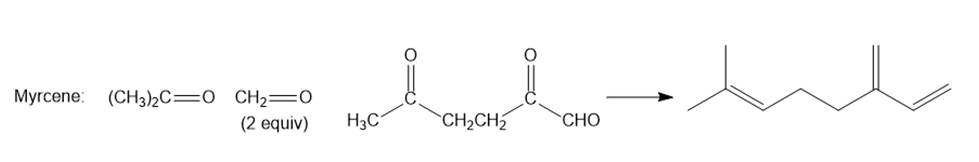
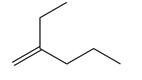
Question: Oximene and myrcene, two hydrocarbons isolated from alfalfa that have the molecular formula , both yield 2,6-dimethyloctane when treated with and a Pd catalyst. Ozonolysis of oximene forms , , and . Ozonolysis of myrcene yields , (two equiv), and . Identify the structures of oximene and myrcene.
Short Answer
Answer