Chapter 12: Q.67. (page 493)
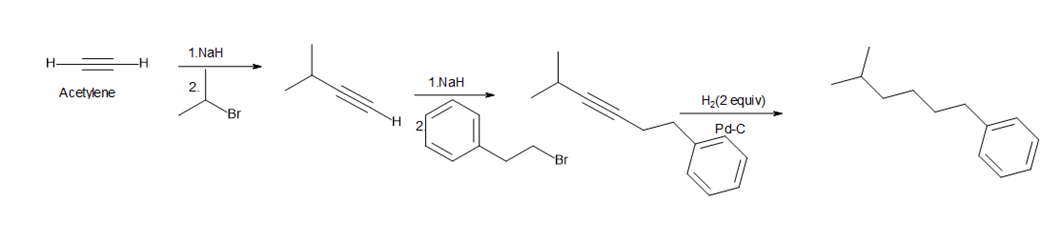
Question:Devise a synthesis of 1-phenyl-5-methylhexane from acetylene, alkylhalides, and any required inorganic reagents.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 12: Q.67. (page 493)
Question:Devise a synthesis of 1-phenyl-5-methylhexane from acetylene, alkylhalides, and any required inorganic reagents.
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Explain why heats of hydrogenation cannot be used to determine the relative stability of 2-methylpent-2-ene and 3-methylpent-1-ene.
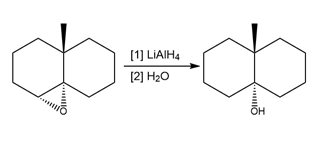
Question: Draw a stepwise mechanism for the reduction of epoxide A to alcohol B using LiAlH4 . What product would be formed if LiAlD4were used as reagent? Indicate the stereochemistry of all stereogenic centers in the product using wedges and dashed wedges.
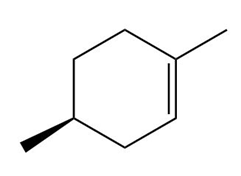
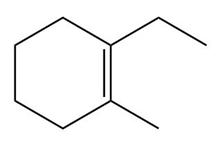
Question: Draw the organic products formed when each alkene is treated with H2 / Pd-C. Indicate the three-dimensional structure of all stereoisomers formed.
a.
b.
c.
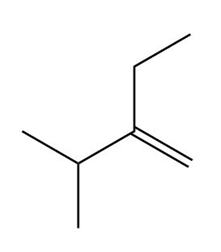
Question: What alkene is needed to synthesize each 1,2-diol using [1] followed by NAHSO3 in H2O ; or [2] CH3CO3H followed by -OH in H2O ?
a.
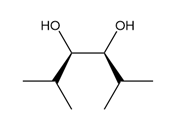
b.
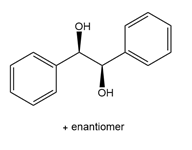
c.
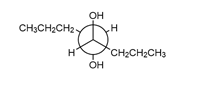
Question: Draw the organic products in each of the following reactions.
a.
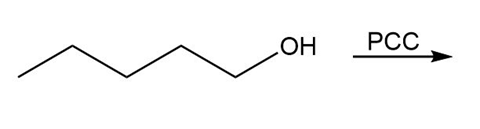
b.
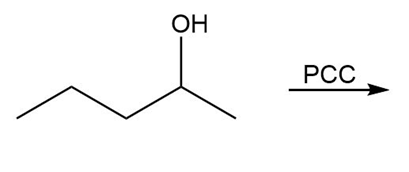
c.
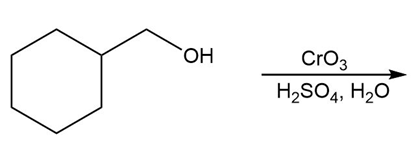
d.
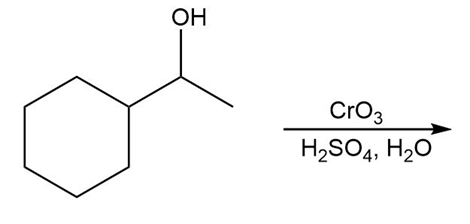
What do you think about this solution?
We value your feedback to improve our textbook solutions.