Chapter 12: PROBLEM 12.44 (page 489)
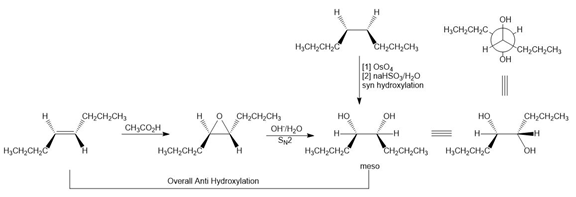
Question: What alkene is needed to synthesize each 1,2-diol using [1] followed by NAHSO3 in H2O ; or [2] CH3CO3H followed by -OH in H2O ?
a.
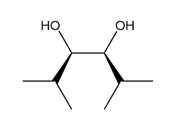
b.
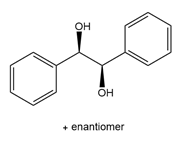
c.
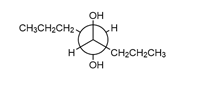
Short Answer
Answer
a.
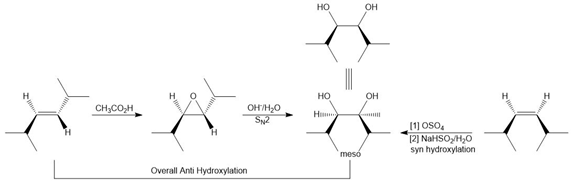
b.
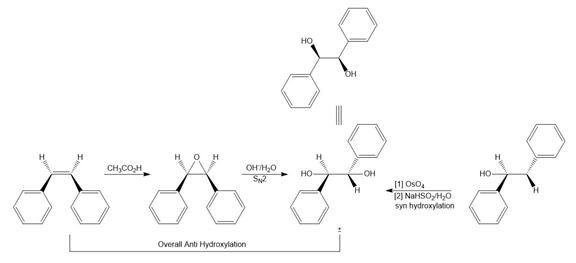
c.
Learning Materials
Features
Discover
Chapter 12: PROBLEM 12.44 (page 489)
Question: What alkene is needed to synthesize each 1,2-diol using [1] followed by NAHSO3 in H2O ; or [2] CH3CO3H followed by -OH in H2O ?
a.

b.

c.

Answer
a.

b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Draw the organic products formed when each alkene is treated with H2 / Pd-C. Indicate the three-dimensional structure of all stereoisomers formed.
a.
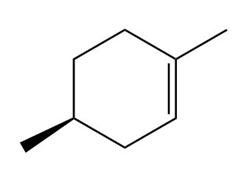
b.
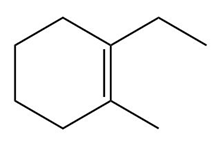
c.
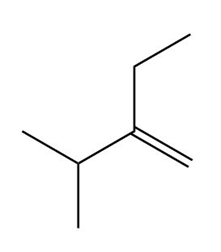
Question:Devise a synthesis of (3R,4S)-3,4-dichlorohexane from acetylene and any needed organic compounds or inorganic reagents.
Question: Draw all alkenes that react with one equivalent of H2 in the presence of a palladium catalyst to form each alkane. Consider constitutional isomers only
a.
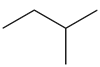
b.
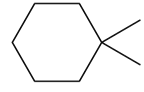
c.
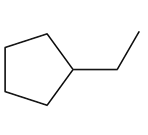
Question: Devise a synthesis of muscalure, the sex pheromone of the common housefly, from acetylene and any other required reagents.
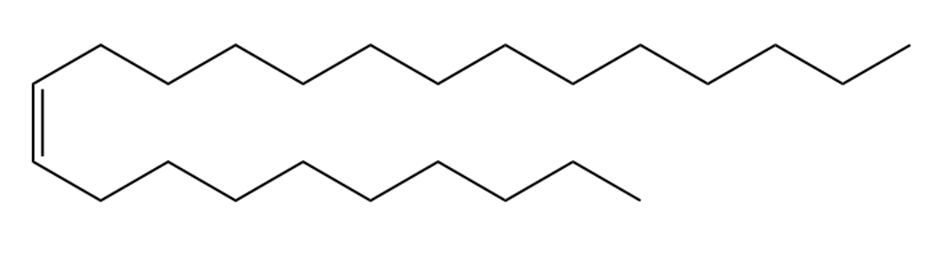
Muscalure
Question: In the oxidation of cyclohexanols, it is generally true that sterically hindered alcohols react faster than unhindered alcohols. Which of the following alcohols should be oxidized more rapidly?
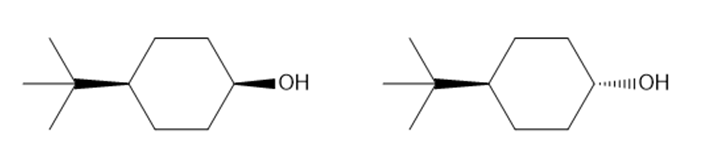
What do you think about this solution?
We value your feedback to improve our textbook solutions.