Chapter 12: Q.71. (page 455)
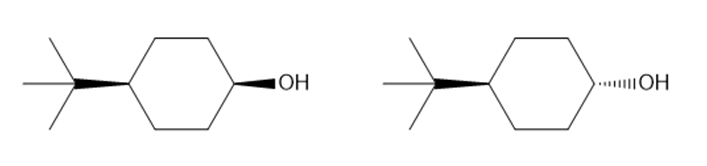
Question: In the oxidation of cyclohexanols, it is generally true that sterically hindered alcohols react faster than unhindered alcohols. Which of the following alcohols should be oxidized more rapidly?
Short Answer
Answer
Cyclohexanol which will react faster will be:
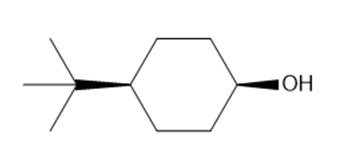
OH group in axial position



