Chapter 12: Q.74. (page 494)
Question: Draw a stepwise mechanism for the following reaction.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 12: Q.74. (page 494)
Question: Draw a stepwise mechanism for the following reaction.
Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Explain why heats of hydrogenation cannot be used to determine the relative stability of 2-methylpent-2-ene and 3-methylpent-1-ene.
Question: Draw the products formed when both cis- and trans-but-2-ene are treated with OsO4, followed by hydrolysis with NaHSO3+H2O. Explain how these reactions illustrate that syndihydroxylation is stereospecific.
Question: Sharpless epoxidation of allylic alcohol X forms compound Y. Treatment of Y with NaOH and in an alcohol–water mixture forms Z. Identify the structure of Y and draw a mechanism for the conversion of Y to Z. Account for the stereochemistry of the stereogenic centers in Z. Z has been used as an intermediate in the synthesis of chiral carbohydrates.
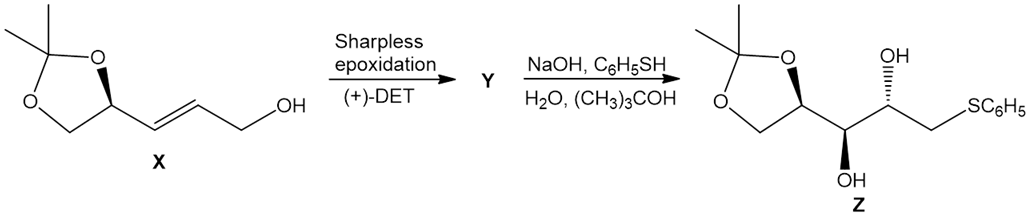
Question: What product is formed when is treated with each reagent: (a) H2 (excess), Pd-C; ; (b) H2(1 equiv), Lindlar catalyst; (c) H2(excess), Lindlar catalyst; (d) Na,NH3 ?
Question: Draw the compounds formed when triacylglycerol A is treated with each reagent, forming compounds B and C. Rank A, B, and C in order of increasing melting point.
A.
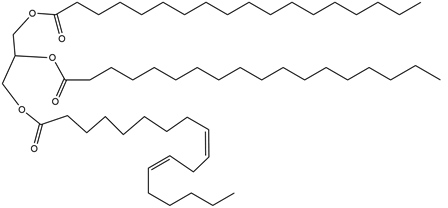
a. (excess), Pd-C (Compound B)
b. (1 equiv), Pd-C (Compound C)
What do you think about this solution?
We value your feedback to improve our textbook solutions.