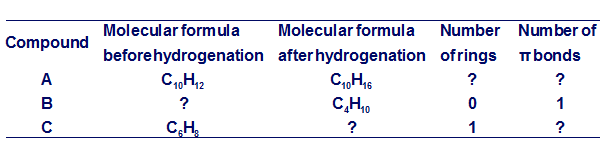
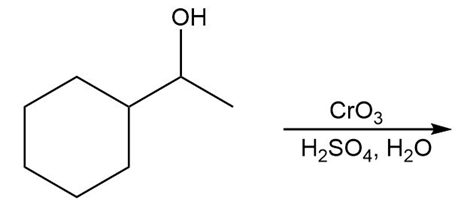
Chapter 12: PROBLEM 12.8 (page 463)
Question: Draw the compounds formed when triacylglycerol A is treated with each reagent, forming compounds B and C. Rank A, B, and C in order of increasing melting point.
A.
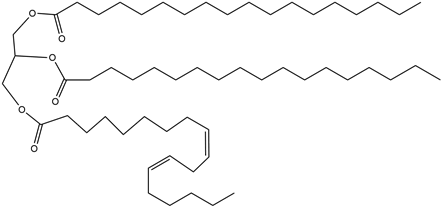
a. (excess), Pd-C (Compound B)
b. (1 equiv), Pd-C (Compound C)
Short Answer
Answer
Compound B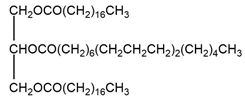
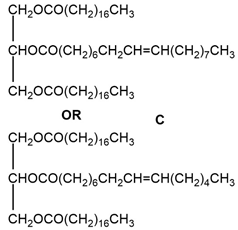
Compound C
 Reaction in excess of catalyst
Reaction in excess of catalyst The reaction using only one equivalence of catalyst
The reaction using only one equivalence of catalyst