Chapter 12: PROBLEM 12.19 (page 475)
Question: Draw the products formed when each alkene is treated with O3 followed by Zn, H2O.
a.
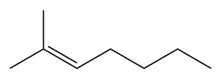
b.
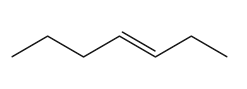
c.
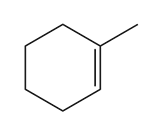
Short Answer
Answer
a.
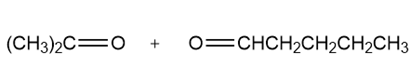
b.
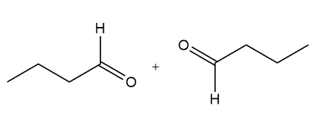
c.
Learning Materials
Features
Discover
Chapter 12: PROBLEM 12.19 (page 475)
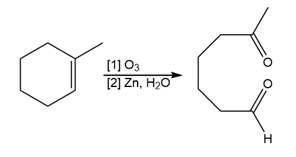
Question: Draw the products formed when each alkene is treated with O3 followed by Zn, H2O.
a.

b.

c.

Answer
a.
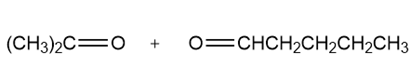
b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Draw the products formed when both cis- and trans-but-2-ene are treated with a peroxyacid followed by – OH (in H2O ). Explain how these reactions illustrate that anti-dihydroxylation is stereospecific.
Question: It is sometimes necessary to isomerize a cis alkene to a trans alkene in a synthesis, a process that cannot be accomplished in a single step. Using the reactions that you have learned in Chapters 8–12, devise a stepwise method to convert cis-but-2-ene to trans-but-2-ene.
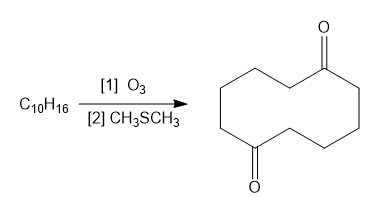
Question: What alkene or alkyne yields each set of products after oxidative cleavage with ozone?
a.
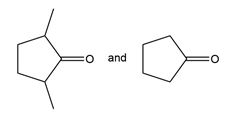
b.
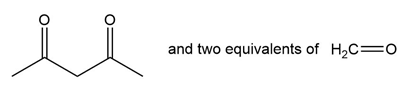
c.
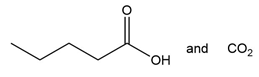
d.
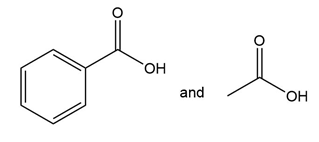
Question: Identify the starting material in each reaction.
a.
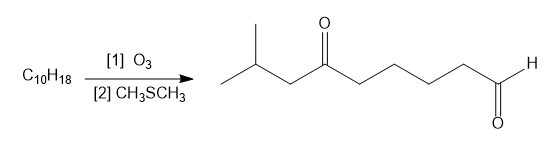
b.
Question: Oximene and myrcene, two hydrocarbons isolated from alfalfa that have the molecular formula , both yield 2,6-dimethyloctane when treated with and a Pd catalyst. Ozonolysis of oximene forms , , and . Ozonolysis of myrcene yields , (two equiv), and . Identify the structures of oximene and myrcene.
What do you think about this solution?
We value your feedback to improve our textbook solutions.