Chapter 12: PROBLEM 12.16 (page 470)
Question: Draw all stereoisomers formed when each alkene is treated with mCPBA.
a.
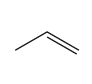
b.
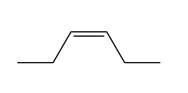
c.
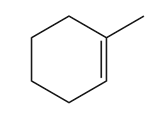
Short Answer
Answer
a.
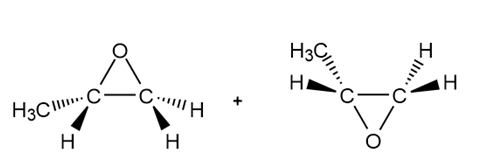
b.
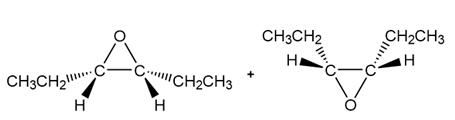
c.
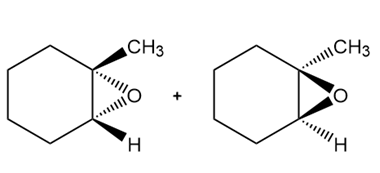
Learning Materials
Features
Discover
Chapter 12: PROBLEM 12.16 (page 470)
Question: Draw all stereoisomers formed when each alkene is treated with mCPBA.
a.

b.

c.

Answer
a.

b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
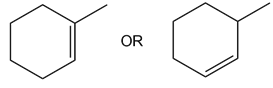
Question: Which alkene in each pair has the larger Heat of hydrogenation?
a.
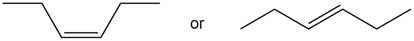
b.
Question: Identify A in the following reaction sequence and draw a mechanism for the conversion of A to B. B has been converted to (S,S)-reboxetine, an antidepressant marketed outside the United States.
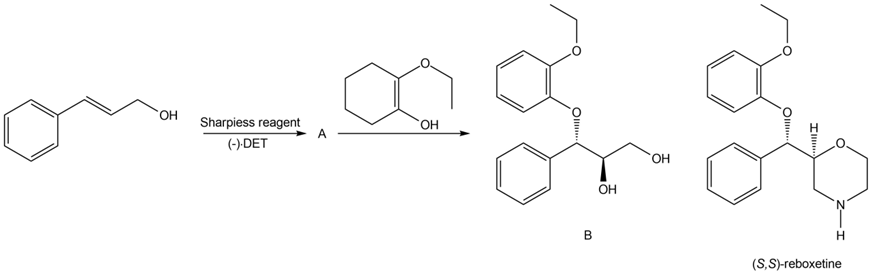
Question: Draw the organic products formed when each alkene is treated with H2 / Pd-C. Indicate the three-dimensional structure of all stereoisomers formed.
a.
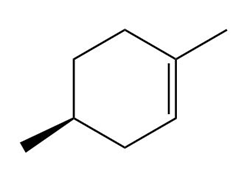
b.
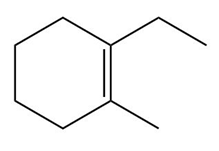
c.
Question: In the oxidation of cyclohexanols, it is generally true that sterically hindered alcohols react faster than unhindered alcohols. Which of the following alcohols should be oxidized more rapidly?
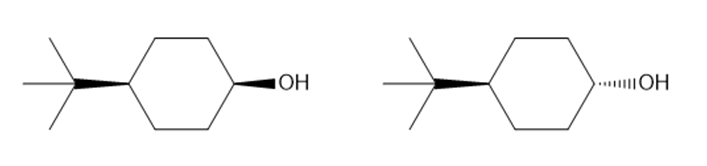
Question: What allylic alcohol and DET isomer are needed to make each chiral epoxide using a sharpless asymmetric epoxidation reaction?
What do you think about this solution?
We value your feedback to improve our textbook solutions.