Chapter 12: PROBLEM 12.15 (page 469)
Question:What epoxide is formed when each alkene is treated with mCPBA?
a.
b.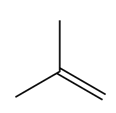
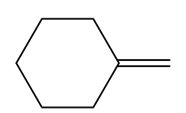
c.
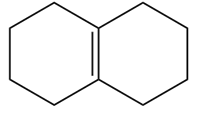
Short Answer
Answer
a.
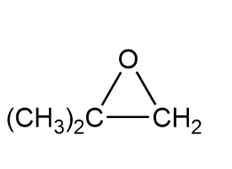
b.
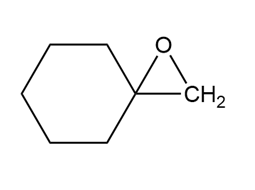
c.
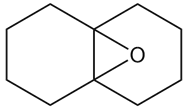
Learning Materials
Features
Discover
Chapter 12: PROBLEM 12.15 (page 469)
Question:What epoxide is formed when each alkene is treated with mCPBA?
a.
b.

c.

Answer
a.

b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
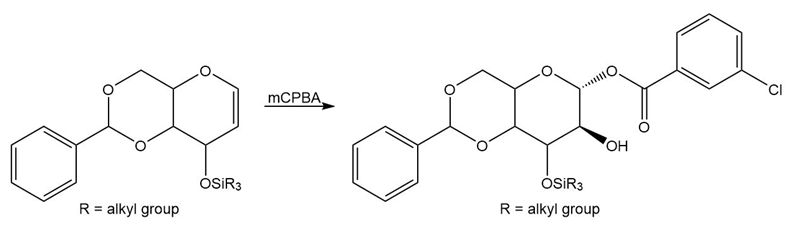
Question: Draw a stepwise mechanism for the following reaction.
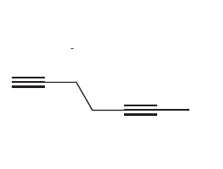
Question: Draw the products formed when each alkyne is treated with O3 followed by H2O .
a.
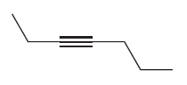
b.
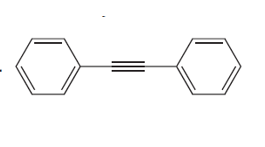
c.
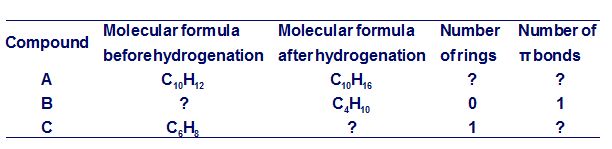
Question: Complete the missing information for compounds A, B, and C, each subjected to hydrogenation. The number of rings and π bonds refers to the reactant (A, B, or C) prior to hydrogenation.
Question: Draw the products formed when both cis- and trans-but-2-ene are treated with a peroxyacid followed by – OH (in H2O ). Explain how these reactions illustrate that anti-dihydroxylation is stereospecific.
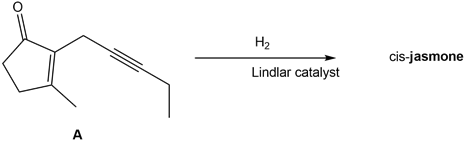
Question: What is the structure of cis-jasmone, a natural product isolated from jasmine flowers, formed by the treatment of alkyne A with H2in the presence of the Lindlar catalyst?
What do you think about this solution?
We value your feedback to improve our textbook solutions.