Chapter 26: Q 16. (page 1066)
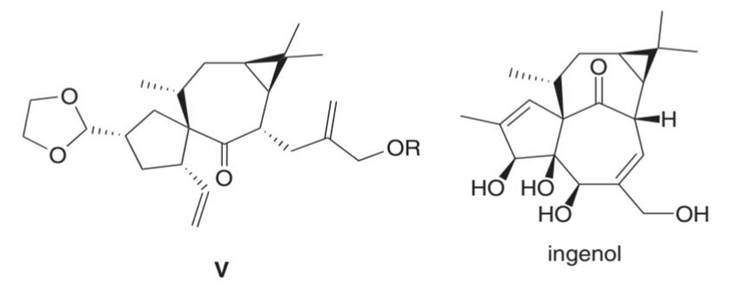
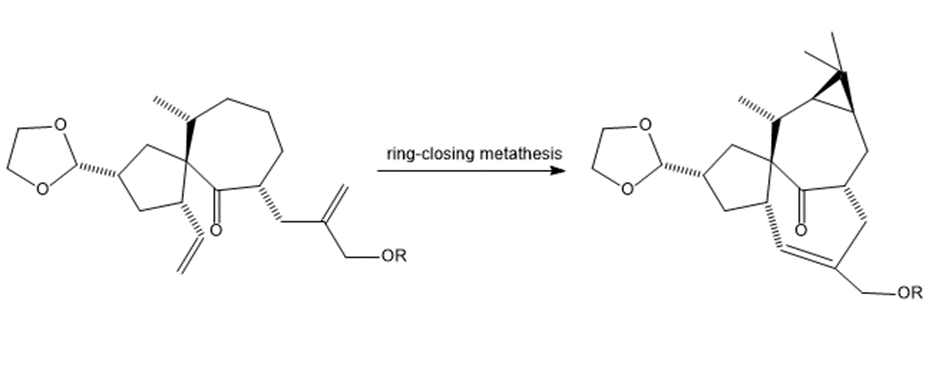
What product is formed by ring-closing metathesis of compound V, a key intermediate in the synthesis of ingenol, a natural product mentioned in the chapter opener?
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 26: Q 16. (page 1066)
What product is formed by ring-closing metathesis of compound V, a key intermediate in the synthesis of ingenol, a natural product mentioned in the chapter opener?

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
What products are formed when cis-pent-2-ene undergoes metathesis? Use this reaction to explain why metathesis of a 1, 2-disubstituted alkene is generally not a practical method for alkene synthesis?
What steps are needed to convert but-1-ene \((C{H_3}C{H_2}CH = C{H_2})\) to octane \((C{H_3}{(C{H_2})_6}C{H_3})\)using a coupling reaction with an organocuprate reagent? All carbon atoms in octane mustcome from but-1-ene.
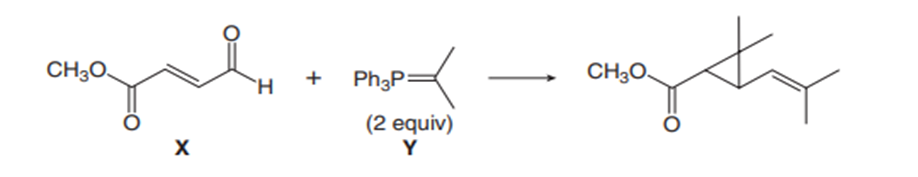
Dimethyl cyclopropanes can be prepared by the reaction of an α, β-unsaturated carbonyl compound X with two equivalents of a Wittig reagent Y. Draw a stepwise mechanism for this reaction.
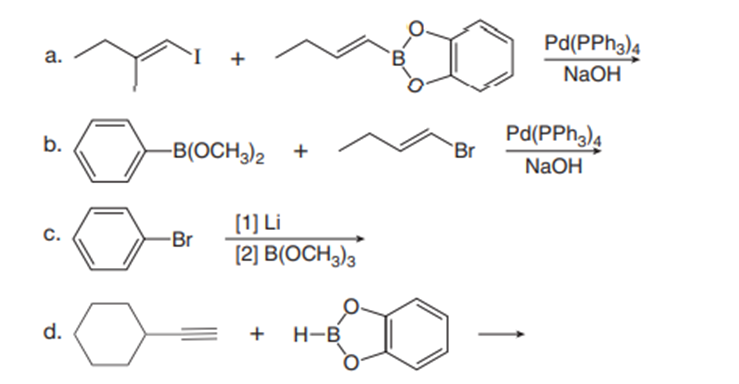
Draw the product of each reaction.
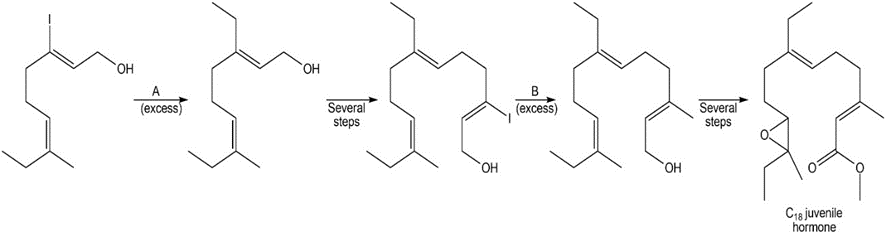
Identify reagents A and B in the following reaction scheme. This synthetic sequence was used to prepare the C18 juvenile hormone (Figure 20.6).
What do you think about this solution?
We value your feedback to improve our textbook solutions.