Chapter 26: Q 2. (page 1051)
Identify reagents A and B in the following reaction scheme. This synthetic sequence was used to prepare the C18 juvenile hormone (Figure 20.6).

Short Answer
Answer

Learning Materials
Features
Discover
Chapter 26: Q 2. (page 1051)
Identify reagents A and B in the following reaction scheme. This synthetic sequence was used to prepare the C18 juvenile hormone (Figure 20.6).

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
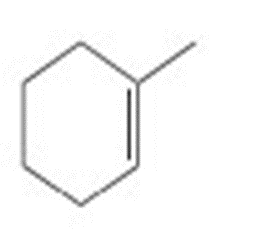
Devise a synthesis of each compound from cyclohexene and any required organic or inorganic reagents.
a.
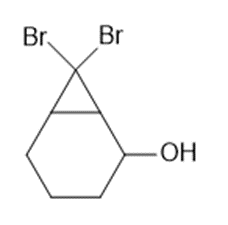
b.

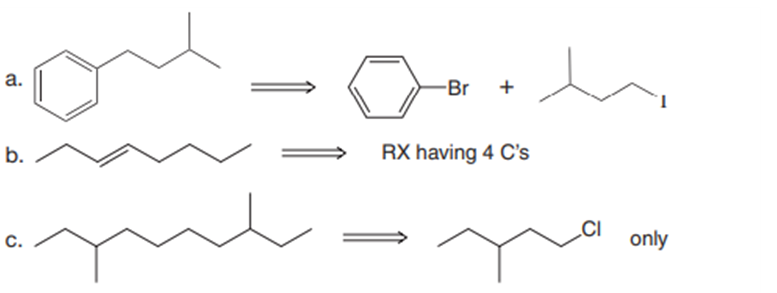
Synthesize each product from the given starting materials using an organocuprate coupling reaction.
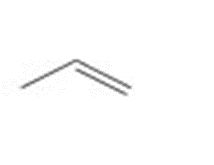
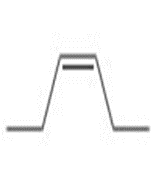
Draw all stereoisomers formed when each alkene is treated with and .
a.
b.
c.
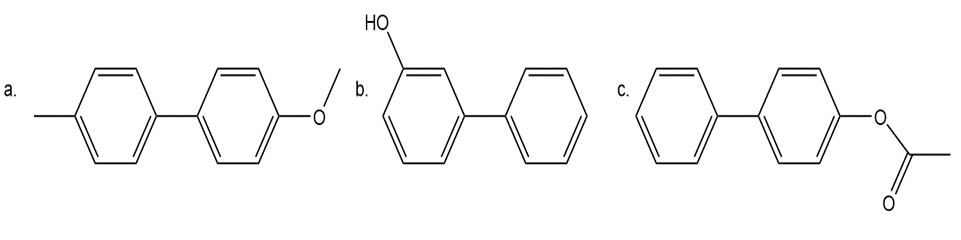
Bi-aryls, compounds containing two aromatic rings joined by a C-C bond, can often be efficiently made by two different Suzuki couplings; that is, either aromatic ring can be used to form the organoborane needed for coupling. In some cases, however, only one route is possible. With this in mind, synthesize each of the following bi-aryls using benzene as the starting material for each aromatic ring. When more than one route is possible, draw both of them. You may use any required organic or inorganic reagents.
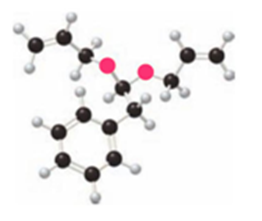
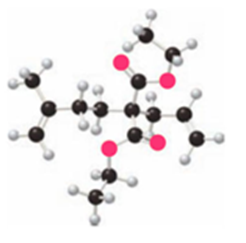
What product is formed by ring-closing metathesis of each compound?
a.
b.
What do you think about this solution?
We value your feedback to improve our textbook solutions.