Chapter 26: Q 9 (page 1061)
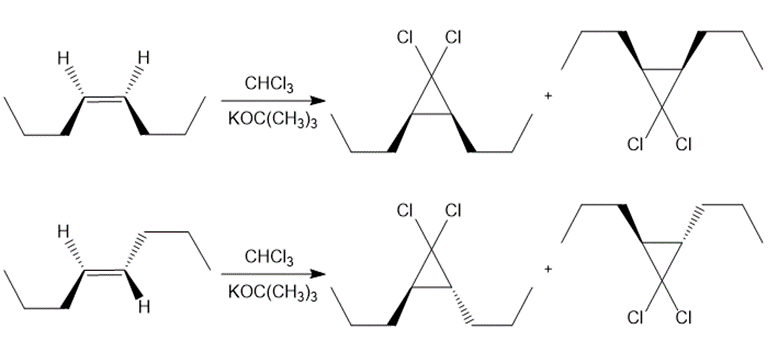
Draw all stereoisomers formed when each alkene is treated with and .
a.
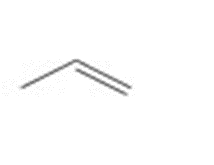
b.
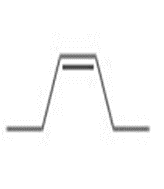
c.
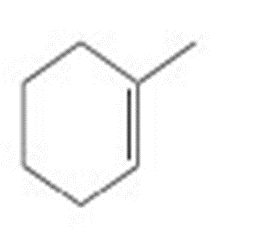
Short Answer
Answer
a.
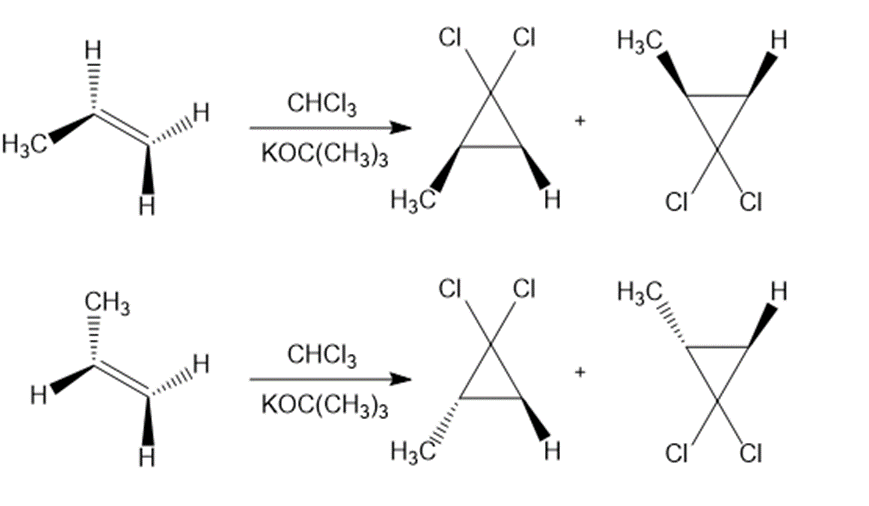
b.
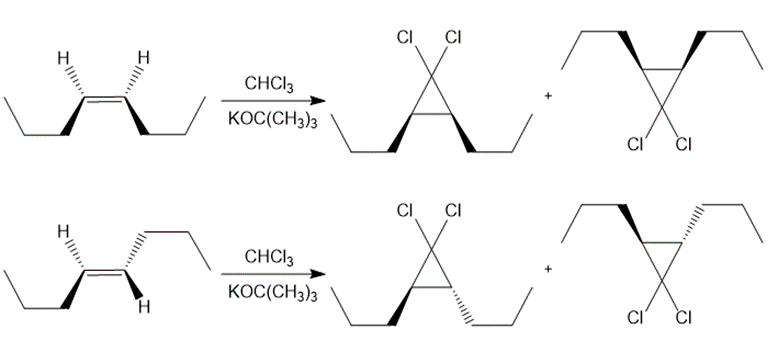
c.
Learning Materials
Features
Discover
Chapter 26: Q 9 (page 1061)
Draw all stereoisomers formed when each alkene is treated with and .
a.

b.

c.

Answer
a.

b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
Metathesis reactions can be carried out with two different alkene substrates in one reaction mixture. Depending on the substitution pattern around the C=C, the reaction may lead to one major product or a mixture of many products. For each pair of alkene substrates, draw all metathesis products formed. (Disregard any starting materials that may also be present at equilibrium.) With reference to the three examples, discuss when alkene metathesis with two different alkenes is a synthetically useful reaction.
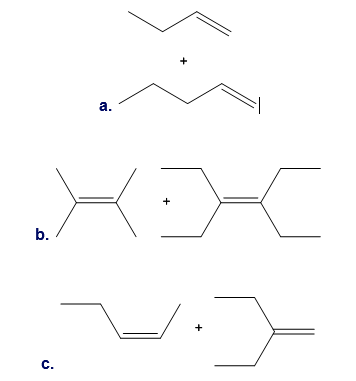
Draw the products formed in each reaction.
a.
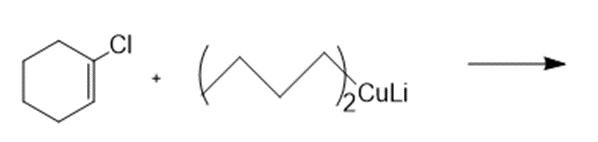
b.
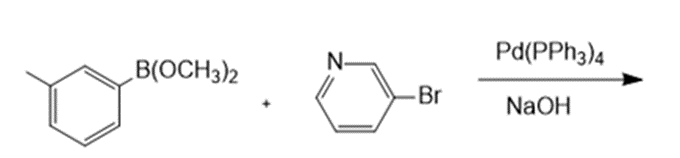
c.
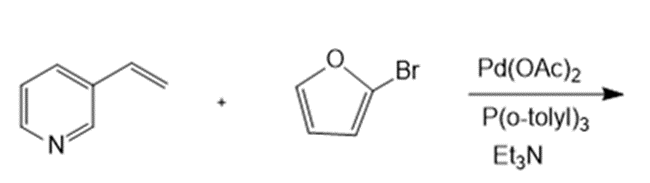
d.
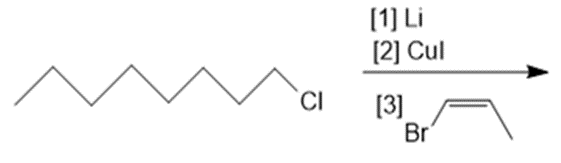
e.
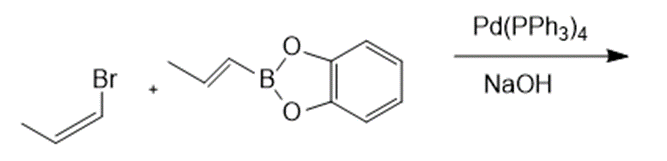
f.
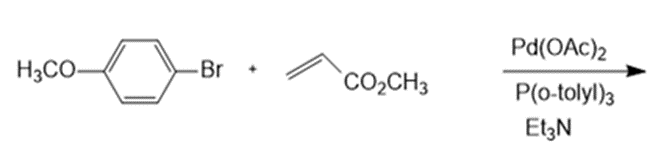
g.
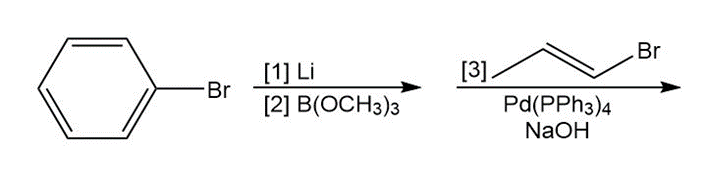
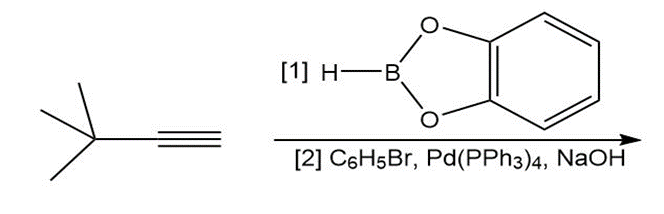
h.
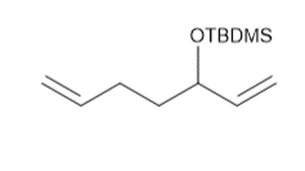
Draw the product formed from the ring-closing metathesis of each compound. Then, devisea synthesis of each metathesis starting material using any of the following compounds: , alcohols with less than five carbons, and any needed organic and inorganicreagents.
a.

b.
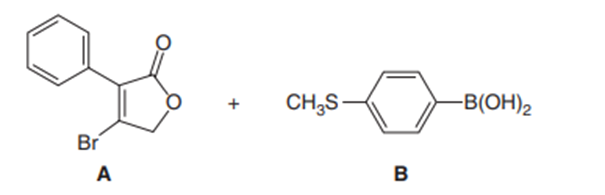
One step in the synthesis of the nonsteroidal anti-inflammatory drug rofecoxib (trade name Vioxx) involves Suzuki coupling of A and B. What product is formed in this reaction?
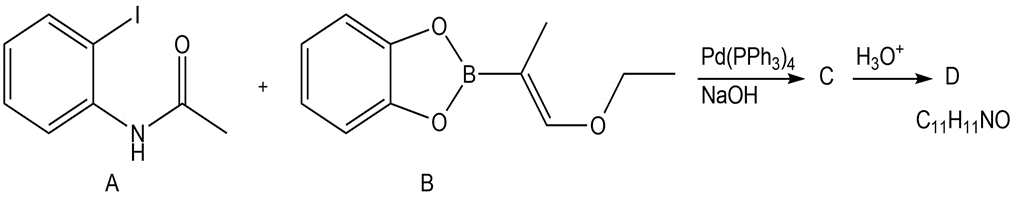
Suzuki coupling of aryl iodide A and vinyl borane B affords compound C, which is converted to D in the presence of aqueous acid. Identify compounds C and D and draw a stepwise mechanism for the conversion of C to D.
What do you think about this solution?
We value your feedback to improve our textbook solutions.