Chapter 26: Q 3. (page 1052)
Synthesize each product from the given starting materials using an organocuprate coupling reaction.
Short Answer
Answer
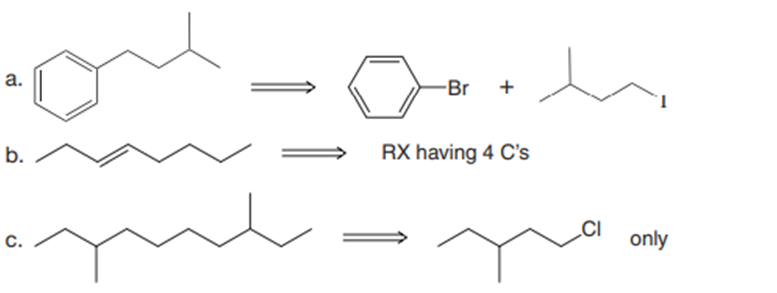
a.
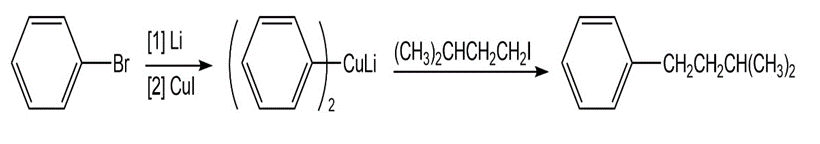
b.
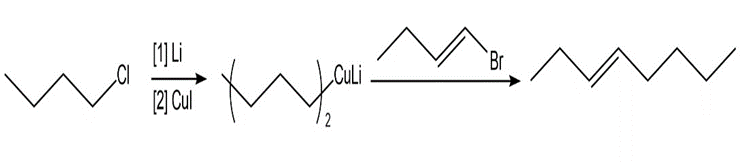
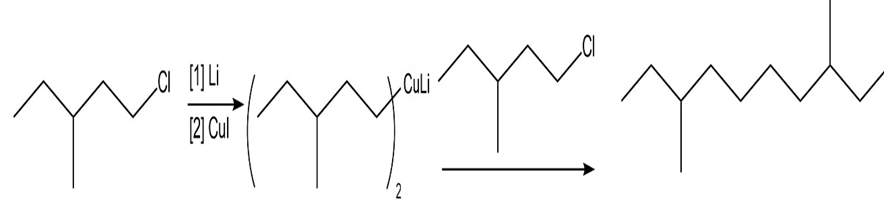
c.
Learning Materials
Features
Discover
Chapter 26: Q 3. (page 1052)
Synthesize each product from the given starting materials using an organocuprate coupling reaction.

Answer
a.
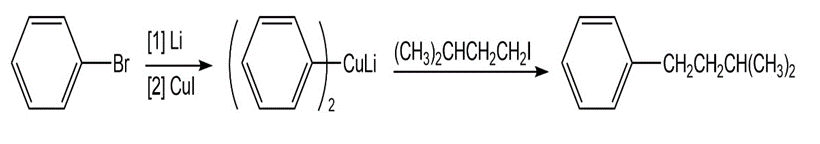
b.
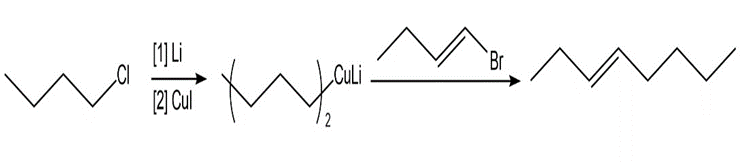
c.

All the tools & learning materials you need for study success - in one app.
Get started for free
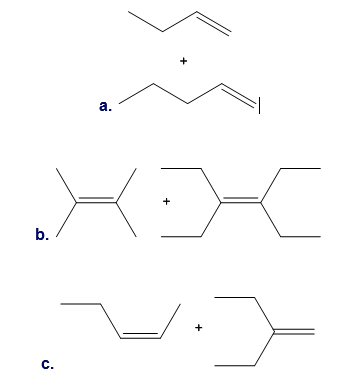
Metathesis reactions can be carried out with two different alkene substrates in one reaction mixture. Depending on the substitution pattern around the C=C, the reaction may lead to one major product or a mixture of many products. For each pair of alkene substrates, draw all metathesis products formed. (Disregard any starting materials that may also be present at equilibrium.) With reference to the three examples, discuss when alkene metathesis with two different alkenes is a synthetically useful reaction.
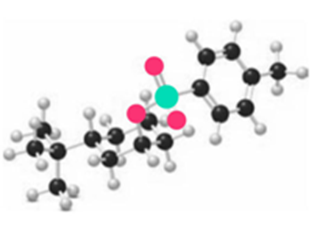
In addition to organic halides, alkyl tosylates (R'OTs, Section 9.13) also react with organocuprates ( ) to form coupling products R-R'. When 2° alkyl tosylates are used as starting materials ( ), inversion of the configuration at a stereogenic center results. Keeping this in mind, draw the product formed when each compound is treated with .
a.
b.
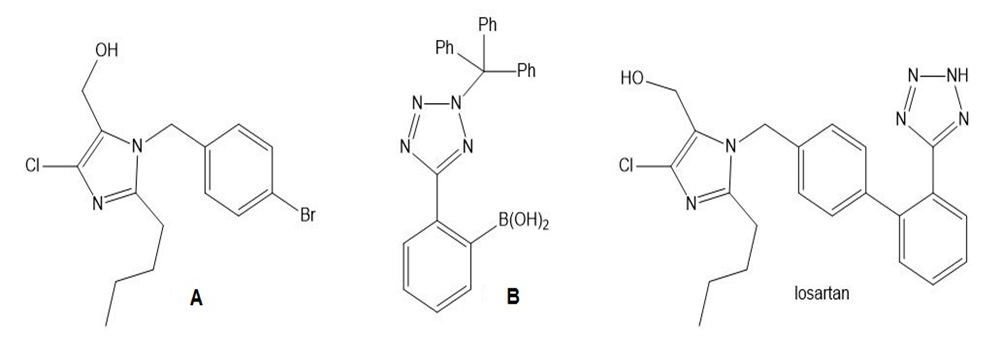
What product is formed in the Suzuki coupling of A and B? This reaction was a key step in the synthesis of losartan, a drug used to treat hypertension.
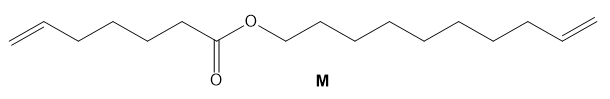
Draw the structure of the two products of molecular formula formed when M is treated with Grubbs catalyst under high-dilution conditions.
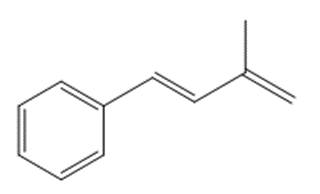
What compound is needed to convert styrene to each product using a Heck reaction?
a.
b.
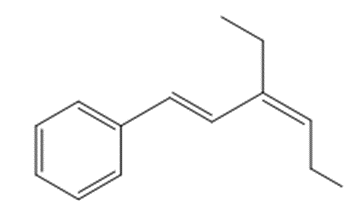
c.
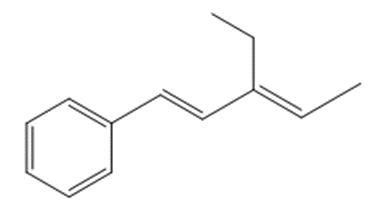
What do you think about this solution?
We value your feedback to improve our textbook solutions.