Chapter 26: Q 23. (page 1069)
What compound is needed to convert styrene to each product using a Heck reaction?
a.
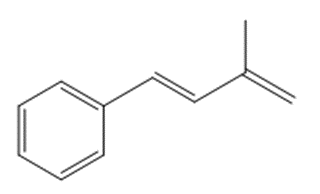
b.
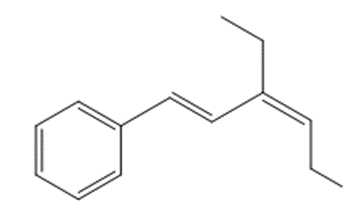
c.
Short Answer
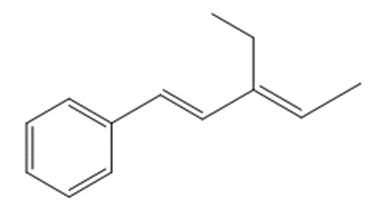
Answer
a.
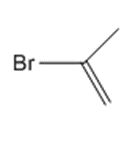
b.
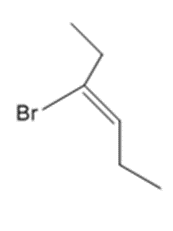
c.
Learning Materials
Features
Discover
Chapter 26: Q 23. (page 1069)
What compound is needed to convert styrene to each product using a Heck reaction?
a.

b.

c.

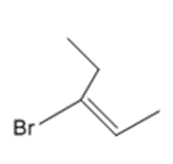
Answer
a.

b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
Devise a synthesis of (E)-1-phenylhex-1-ene ( ) using hydrocarbons having ≤ 6 C’s and a Suzuki reaction as one of the steps.
Devise a synthesis of each substituted cyclopropane. Use acetylene , as a startingmaterial in part (a), and cyclohexanone as a starting material in part (b). You may use anyother organic compounds and any needed reagents.
a.
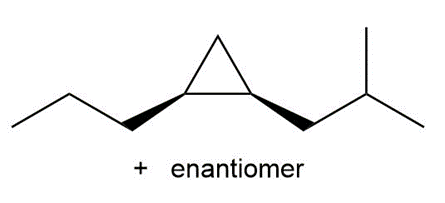
b.
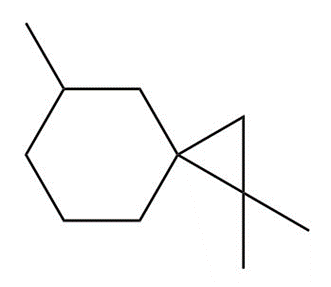
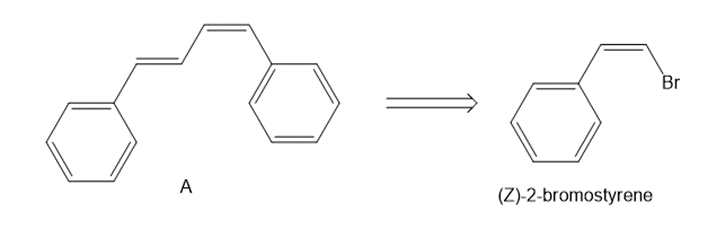
Devise a synthesis of diene A from (Z)-2-bromostyrene as the only organic starting material. Use a Suzuki reaction in one step of the synthesis.
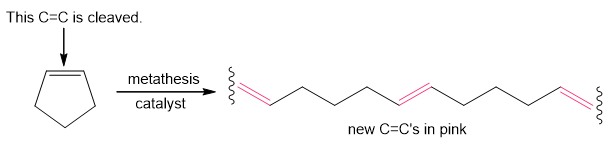
When certain cycloalkenes are used in metathesis reactions, ring-opening metathesis polymerization (ROMP) occurs to form a high molecular weight polymer, as shown with cyclopentene as the starting material. The reaction is driven to completion by relief of strain in the cycloalkene.
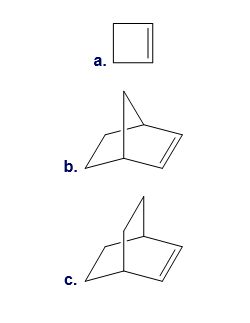
What products are formed by ring-opening metathesis polymerization of each alkene?
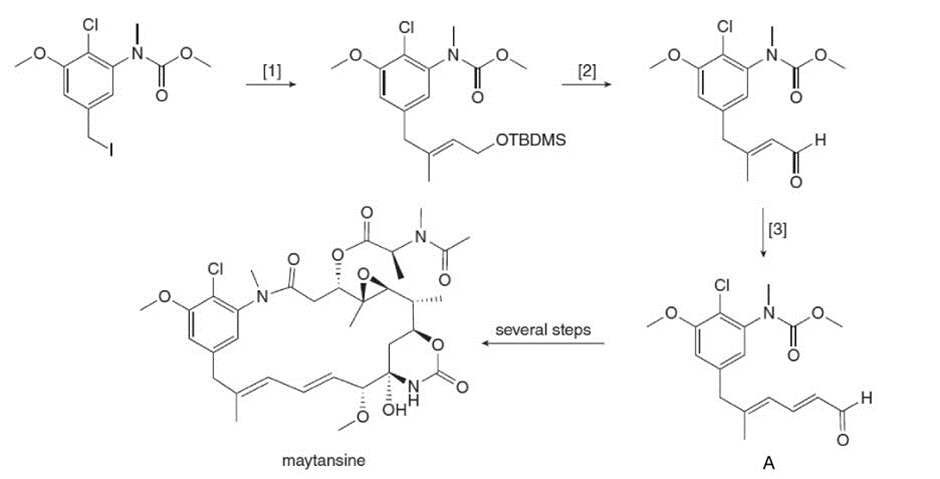
What reagents are needed to carry out transformations [1]–[3] in the synthesis of aldehyde A? A can be converted to the antitumor agent maytansine in several steps.
What do you think about this solution?
We value your feedback to improve our textbook solutions.