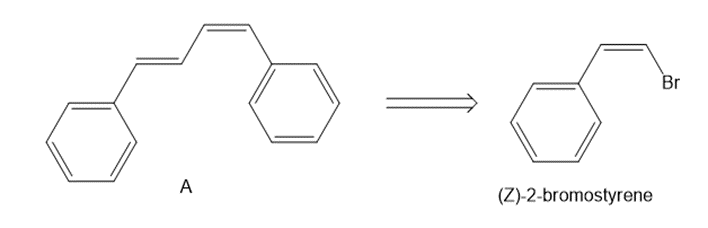
Chapter 26: Q 40. (page 1072)
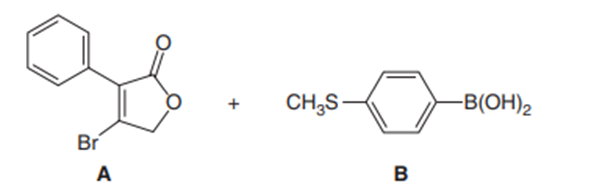
Devise a synthesis of diene A from (Z)-2-bromostyrene as the only organic starting material. Use a Suzuki reaction in one step of the synthesis.
Short Answer
Answer
 Synthesis of compound A
Synthesis of compound A
Learning Materials
Features
Discover
Chapter 26: Q 40. (page 1072)
Devise a synthesis of diene A from (Z)-2-bromostyrene as the only organic starting material. Use a Suzuki reaction in one step of the synthesis.

Answer
 Synthesis of compound A
Synthesis of compound A
All the tools & learning materials you need for study success - in one app.
Get started for free
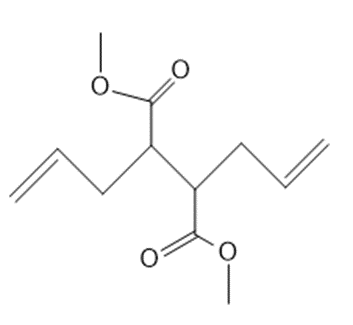
Draw the product formed from ring-closing metathesis of each compound.
a.
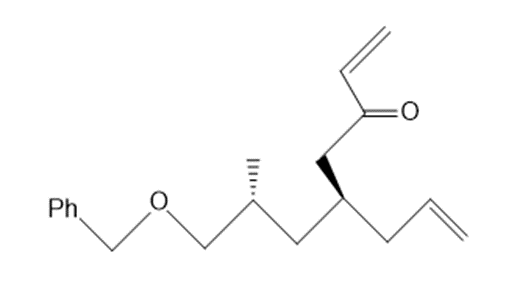
b.
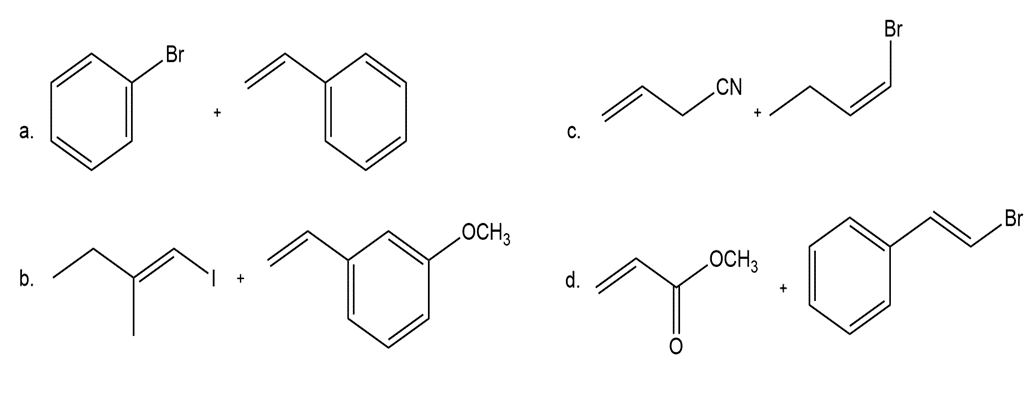
Draw the coupling product formed when each pair of compounds is treated with , , and .
What starting material is needed to prepare each compound by a ring-closing metathesis reaction?
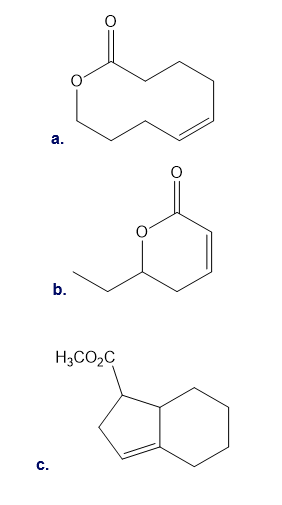
One step in the synthesis of the nonsteroidal anti-inflammatory drug rofecoxib (trade name Vioxx) involves Suzuki coupling of A and B. What product is formed in this reaction?
In addition to using and base to synthesize dihalocarbenes (Section 26.4), dichlorocarbene can be prepared by heating sodium trichloroacetate. Draw a stepwise mechanism for this reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.