Chapter 26: Q 30. (page 1070)
What starting material is needed to prepare each compound by a ring-closing metathesis reaction?

Short Answer
Answer
a.
 Starting material of a
Starting material of a
b.
 Starting material of b
Starting material of b
c.
 Starting material of c
Starting material of c
Learning Materials
Features
Discover
Chapter 26: Q 30. (page 1070)
What starting material is needed to prepare each compound by a ring-closing metathesis reaction?

Answer
a.
 Starting material of a
Starting material of a
b.
 Starting material of b
Starting material of b
c.
 Starting material of c
Starting material of c
All the tools & learning materials you need for study success - in one app.
Get started for free
Treatment of cyclohexene with and Zn(Cu) forms two stereoisomers of molecular formula . Draw their structures and explain why two compounds are formed.
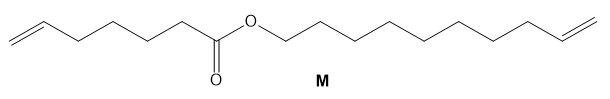
Draw the structure of the two products of molecular formula formed when M is treated with Grubbs catalyst under high-dilution conditions.
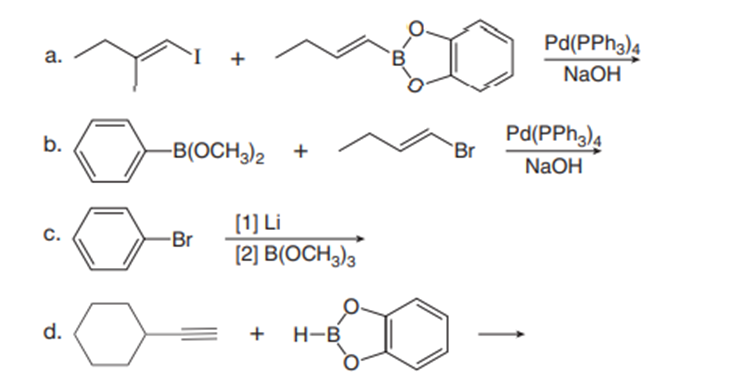
Draw the product of each reaction.
Draw all stereoisomers formed when each alkene is treated with and .
a.
b.

c.
Many variations of ring-closing metathesis have now been reported. Tandem ring-opening– ring-closing metathesis can occur with cyclic alkenes that contain two additional carbon– carbon double bonds. In this reaction, the cycloalkene is cleaved, and two new rings are formed. [1] What compounds are formed in this tandem reaction with the following substrates? [2] Devise a synthesis of the substrate in part (b) that uses a Diels–Alder reaction with diethyl maleate as the dienophile.
a.
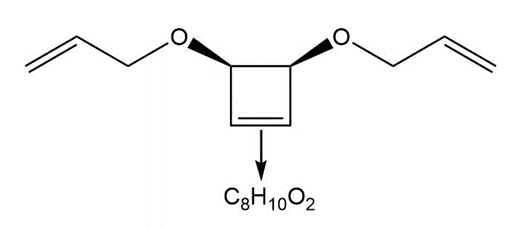
b.
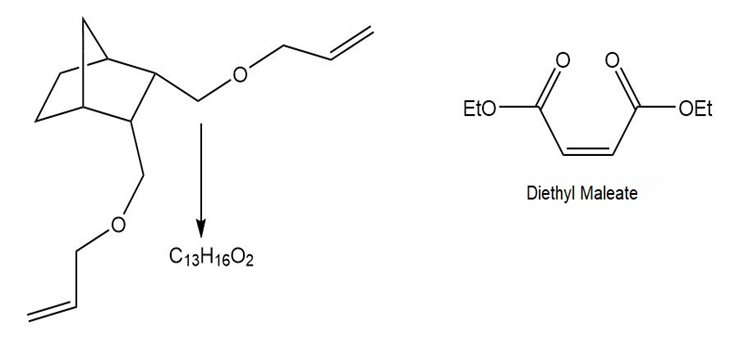
What do you think about this solution?
We value your feedback to improve our textbook solutions.