Chapter 26: Q 28. (page 1070)
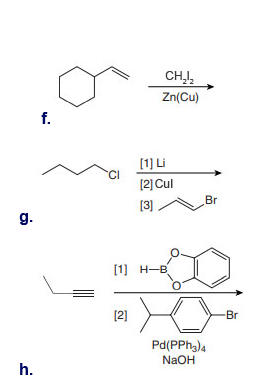
Treatment of cyclohexene with and Zn(Cu) forms two stereoisomers of molecular formula . Draw their structures and explain why two compounds are formed.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 26: Q 28. (page 1070)
Treatment of cyclohexene with and Zn(Cu) forms two stereoisomers of molecular formula . Draw their structures and explain why two compounds are formed.
Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
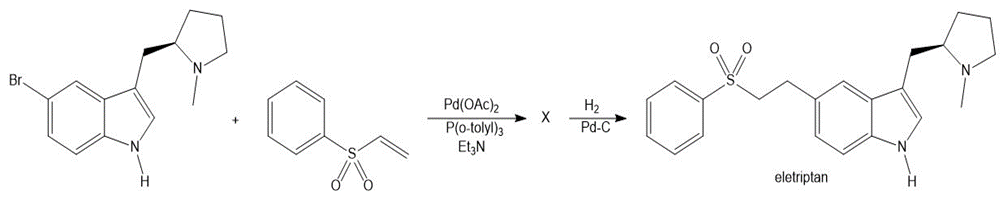
Identify X, an intermediate that was converted to eletriptan (trade name Relpax), a drug used to treat migraines.
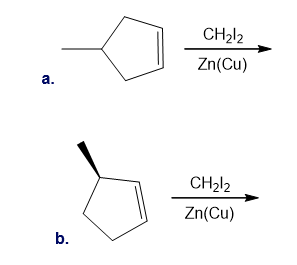
Draw the products (including stereoisomers) formed in each reaction.
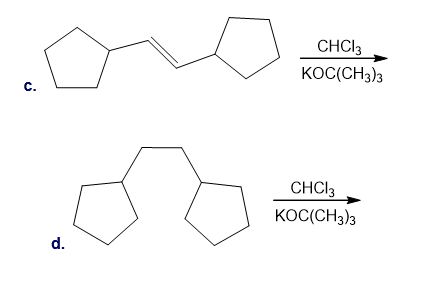
Devise a synthesis of (E)-1-phenylhex-1-ene ( ) using hydrocarbons having ≤ 6 C’s and a Suzuki reaction as one of the steps.
Draw the products formed in each reaction.
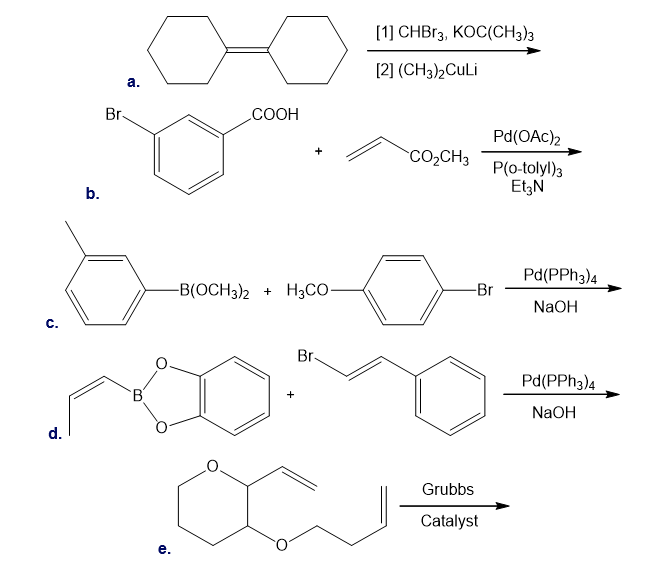
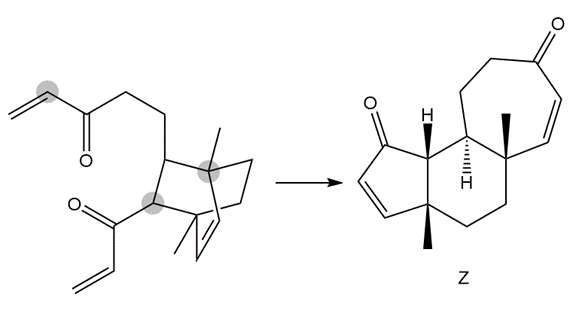
The following conversion, carried out in the presence of Grubbs catalyst and ethylene gas, involves a cascade of metathesis reactions. Draw a reaction sequence that illustrates how the reactant is converted to the product Z, and indicate where each labeled atom in the reactant ends up in Z.
What do you think about this solution?
We value your feedback to improve our textbook solutions.