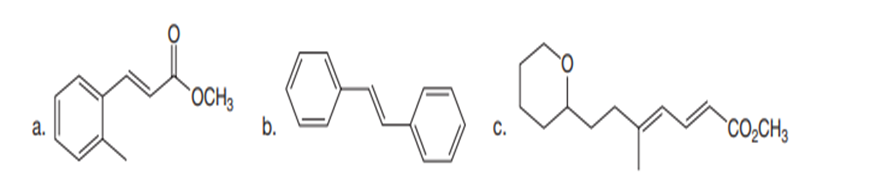
Chapter 26: Q 29. (page 1070)
What ring-closing metathesis product is formed when each substrate is treated with Grubbs catalyst under high-dilution conditions?
a.
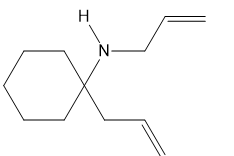
b.
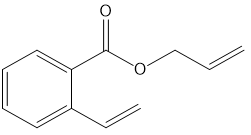
c.
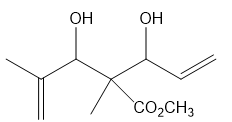
Short Answer
Answer

 Product of b
Product of b
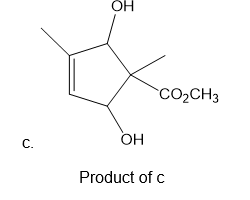
Learning Materials
Features
Discover
Chapter 26: Q 29. (page 1070)
What ring-closing metathesis product is formed when each substrate is treated with Grubbs catalyst under high-dilution conditions?
a.

b.

c.

Answer

 Product of b
Product of b

All the tools & learning materials you need for study success - in one app.
Get started for free
Draw all stereoisomers formed when each alkene is treated with and .
a.

b.
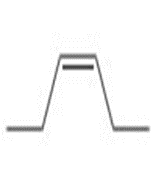
c.
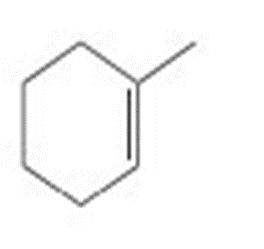
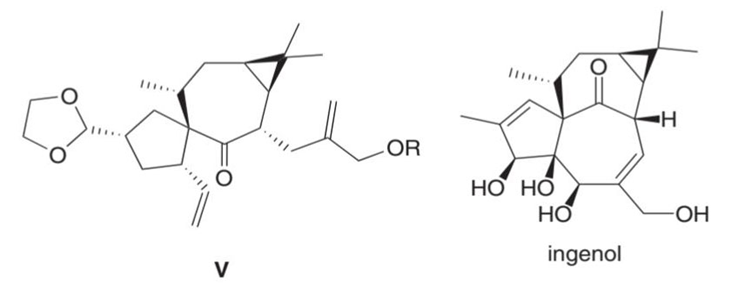
What product is formed by ring-closing metathesis of compound V, a key intermediate in the synthesis of ingenol, a natural product mentioned in the chapter opener?
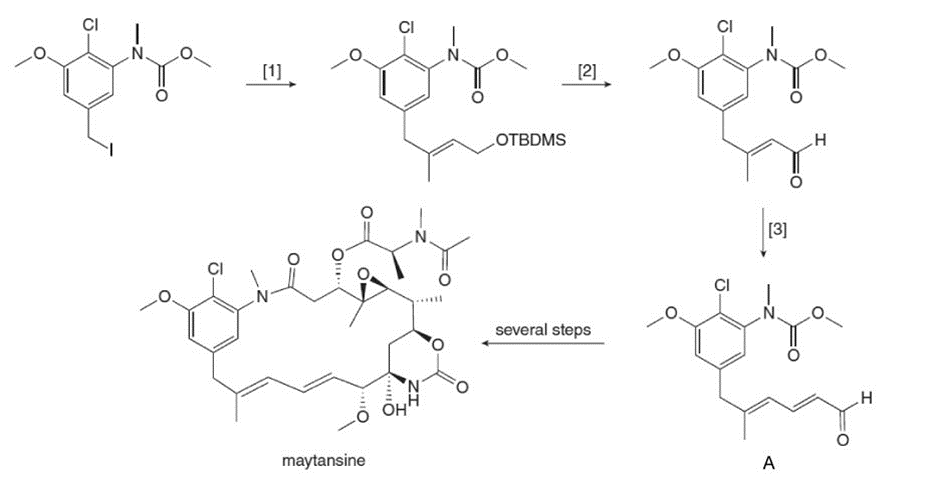
What reagents are needed to carry out transformations [1]–[3] in the synthesis of aldehyde A? A can be converted to the antitumor agent maytansine in several steps.
What starting materials are needed to prepare each compound using a Heck reaction?
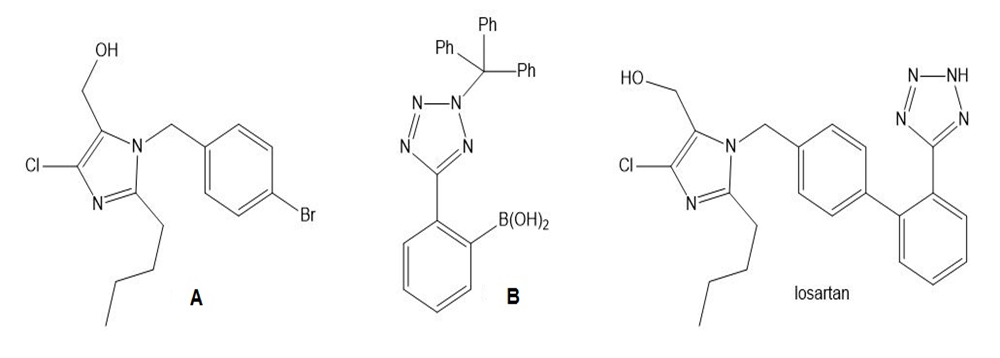
What product is formed in the Suzuki coupling of A and B? This reaction was a key step in the synthesis of losartan, a drug used to treat hypertension.
What do you think about this solution?
We value your feedback to improve our textbook solutions.