Chapter 26: Q 44. (page 1073)
Devise a synthesis of each compound from cyclohexene and any required organic or inorganic reagents.
a.

b.
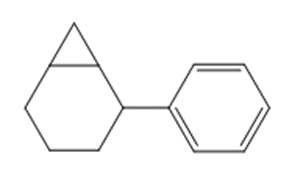
Short Answer
Answer
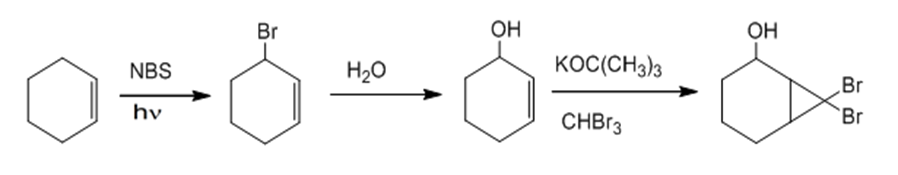
a.
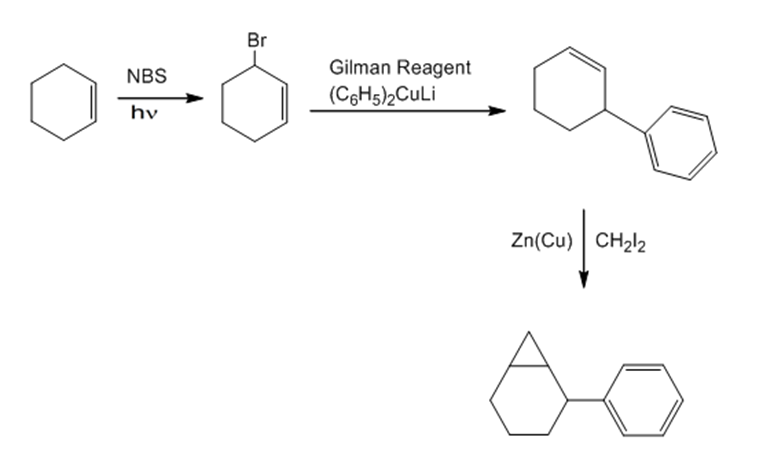
b.
Learning Materials
Features
Discover
Chapter 26: Q 44. (page 1073)
Devise a synthesis of each compound from cyclohexene and any required organic or inorganic reagents.
a.

b.

Answer
a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the product formed from the ring-closing metathesis of each compound. Then, devise a synthesis of each metathesis starting material from benzene, alcohols with less than five carbons, and any needed organic and inorganic reagents.
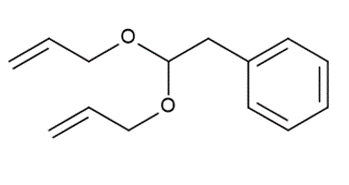
a.

b.
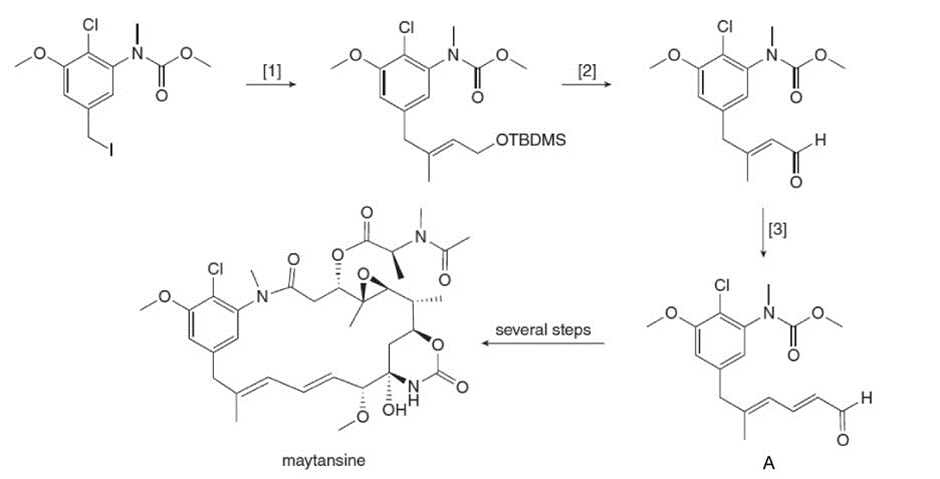
What reagents are needed to carry out transformations [1]–[3] in the synthesis of aldehyde A? A can be converted to the antitumor agent maytansine in several steps.
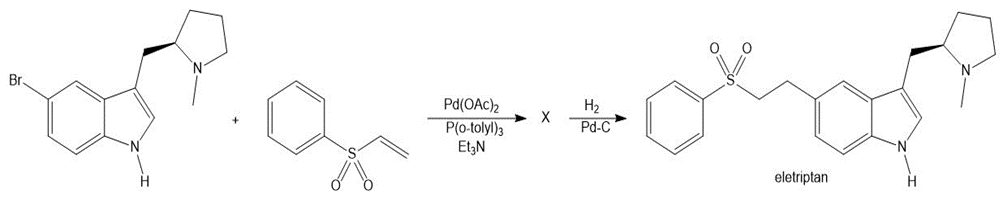
Identify X, an intermediate that was converted to eletriptan (trade name Relpax), a drug used to treat migraines.
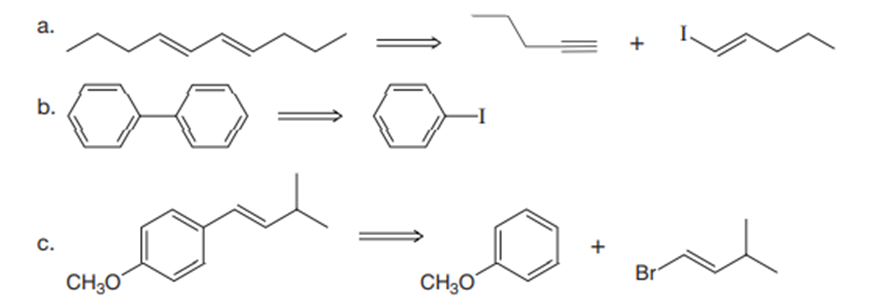
Synthesize each compound from the given starting materials.
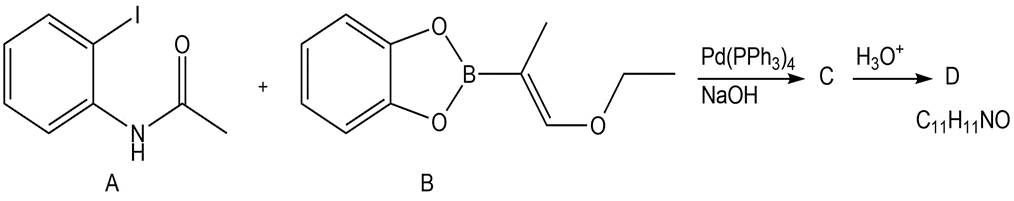
Suzuki coupling of aryl iodide A and vinyl borane B affords compound C, which is converted to D in the presence of aqueous acid. Identify compounds C and D and draw a stepwise mechanism for the conversion of C to D.
What do you think about this solution?
We value your feedback to improve our textbook solutions.