Chapter 26: Q 12. (page 1062)
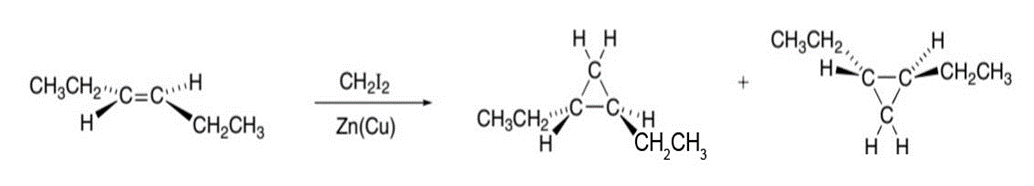
What stereoisomers are formed when trans-hex-3-ene is treated with and Zn(Cu)?
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 26: Q 12. (page 1062)
What stereoisomers are formed when trans-hex-3-ene is treated with and Zn(Cu)?
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
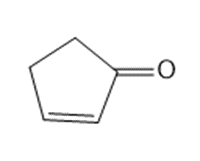
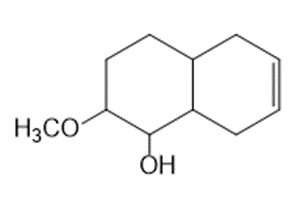
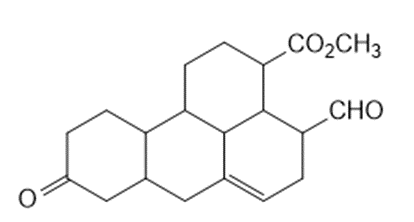
What starting material is needed to synthesize each compound by a ring-closing metathesis reaction?
a.
b.
c.
What ring-closing metathesis product is formed when each substrate is treated with Grubbs catalyst under high-dilution conditions?
a.
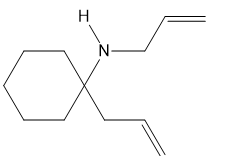
b.
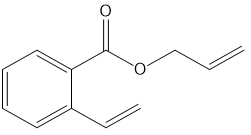
c.
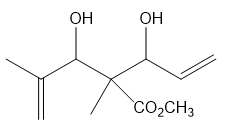
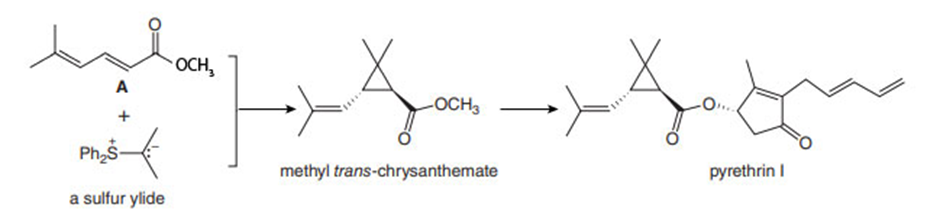
Sulfur ylides, like the phosphorus ylides of Chapter 21, are useful intermediates in organic synthesis. Methyl trans-chrysanthemate, an intermediate in the synthesis of the insecticide pyrethrin I (Section 26.4), can be prepared from diene A and a sulfur ylide. Draw a stepwise mechanism for this reaction.
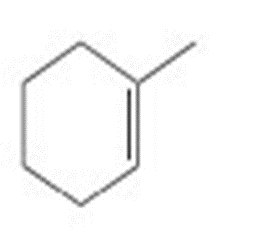
Draw all stereoisomers formed when each alkene is treated with and .
a.
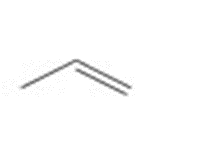
b.
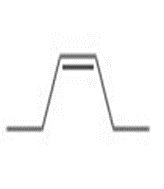
c.
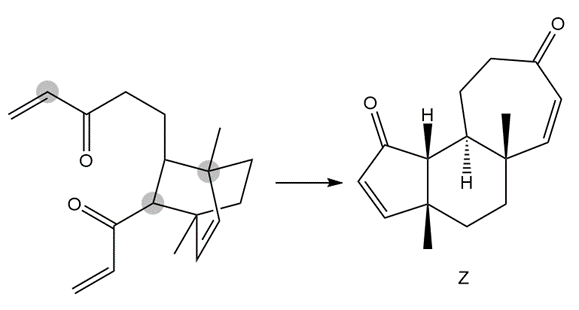
The following conversion, carried out in the presence of Grubbs catalyst and ethylene gas, involves a cascade of metathesis reactions. Draw a reaction sequence that illustrates how the reactant is converted to the product Z, and indicate where each labeled atom in the reactant ends up in Z.
What do you think about this solution?
We value your feedback to improve our textbook solutions.